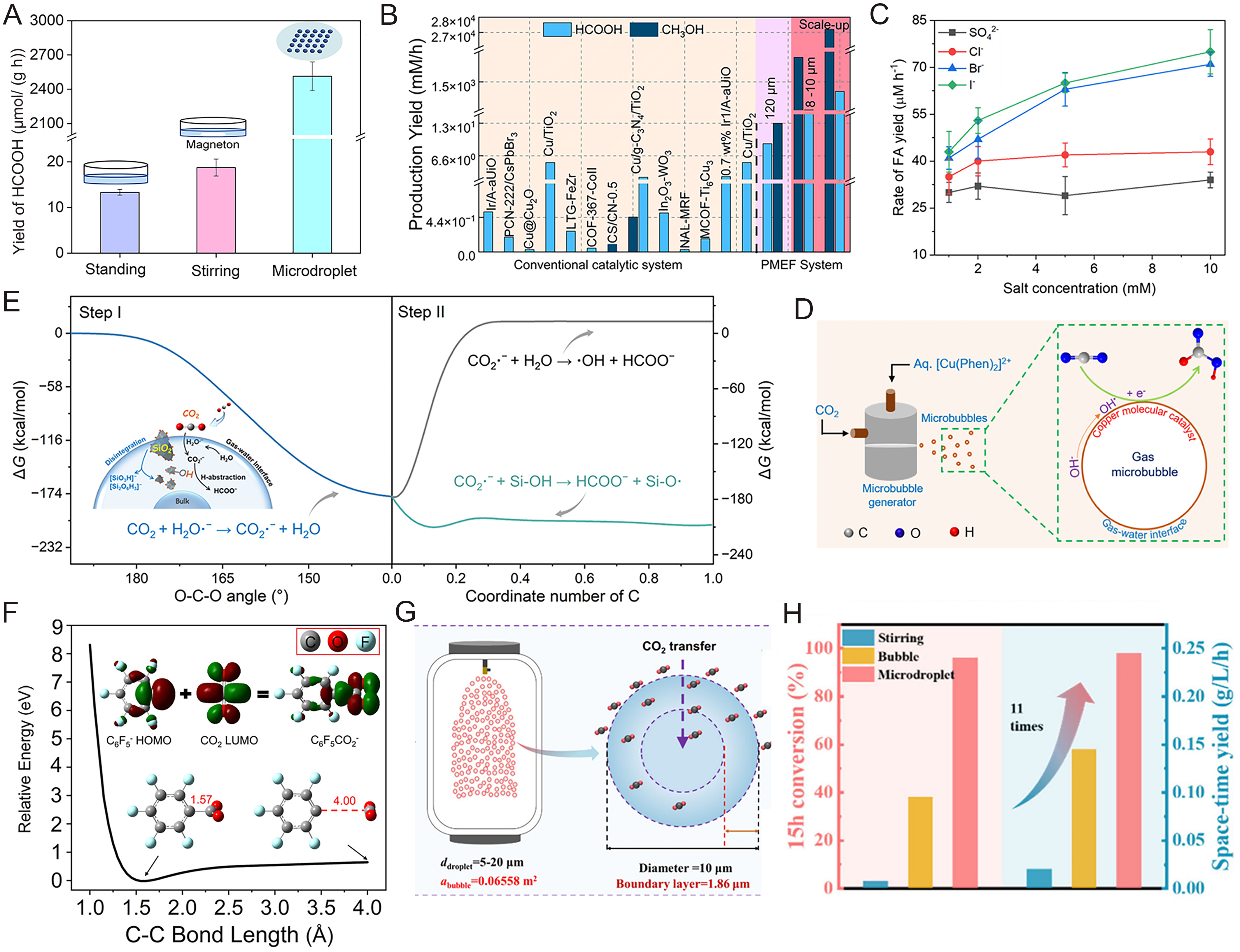
fig2
Figure 2. CO2 activation and conversion in aqueous microdroplets. (A) WO3·0.33H2O photocatalytic CO2 reduction[5]; (B) Comparisons between the yield of CO2 photoreduction in the microdroplet system (PMEF) and the previous observation in bulk solution[1]; (C) Formation of FA (HCOOH) depending on different conditions of the copper(II)-phenanthroline complex [Cu(Phen)2]2+[20]; (D) Schematic of the experimental setup for the reduction of CO2 in microbubbles[20]; (E) SiO2-driven CO2 conversion at the gas-water interface[21]; (F) Fixation of CO2 by C6F5- in microdroplets[6]; (G) Schematic diagram of scale-up reactor and CO2 transfer in microdroplet[15]; (H) Kinetics analysis of CO2 conversion in different scale-up reactors (stirring reactor, bubble reactor, and microdroplet reactor)[15]. Figure 2A is reprinted with permission from Ref.[5], Copyright © 2023 WILEY. Figure 2B is reprinted with permission from Ref.[1], available under a CC BY-NC 4.0. Figure 2C and D is reprinted with permission from Ref.[20], Copyright © 2025 American Chemical Society. Figure 2E is reprinted with permission from Ref.[21], Copyright © 2025 American Chemical Society. Figure 2F is reprinted with permission from Ref.[6], Copyright © 2023 American Chemical Society. Figure 2G and H is reprinted with permission from Ref.[15], Copyright © 2025 American Chemical Society. HOMO: Highest occupied molecular orbital; LUMO: lowest unoccupied molecular orbital.