Liquid metal enabled metallurgy for high-entropy alloys
Abstract
High-entropy alloys (HEAs) have been recognized as a novel class of materials with significant potential in both science and technology. Conventional synthesis of HEAs often requires high-temperature and energy-intensive conditions, limiting scalability and material diversity. Liquid metal metallurgy offers an alternative route for constructing HEA systems under ambient or near-ambient conditions. In this strategy, Ga-, Bi-, and In-based liquid metal systems act as intermediate media that enable multicomponent alloying through relatively low-energy processes. Their fluidic nature supports efficient mixing and mass transport, provides a tunable reaction environment, and facilitates integration with soft matrices, thereby expanding the accessible design space and functional scope. This perspective summarizes recent progress in room-temperature liquid metals and discusses their role in enabling HEA construction via liquid metal-enabled routes. We further present a systematic blueprint covering material selection, processing strategies, and compositional design, and discuss key scientific challenges, including phase control, interfacial chemistry, and property prediction across liquid-to-solid transitions. Finally, we outline future directions, such as artificial intelligence-guided alloy discovery, interfacial reaction modeling, and emerging applications in smart materials, catalysis, and biocompatible electronics.
Keywords
INTRODUCTION
Metallurgy, one of the oldest scientific disciplines, enabled the creation of metallic materials with desirable properties by combining metal elements. With the development of metallurgy, high-entropy alloys (HEAs), which are composed of five or more elements, have garnered attention for their extraordinary properties such as hardness, ductility, and damage tolerance[1]. Unlike conventional alloys, which typically contain no more than three primary elements, HEAs have higher concentrations of each component, usually 5%-35% by atomic ratio, and are also known as multiprincipal element alloys (MPEAs)[2]. Owing to their complex atomic composition and microstructure, HEAs defy conventional alloy prediction rules. This has prompted the development of new physical metallurgy theories to explain their unique properties and underlying mechanisms, leading to oriented synthesis strategies and atomic-level characterization methods, and endowing them with exceptional properties that conventional alloys can hardly achieve[3].
Despite their compositional complexity, many HEAs crystallize into simple solid-solution phases, rather than forming numerous complex intermetallic compounds (IMCs). This behavior is attributed to the high entropy of the alloy system, which reflects the randomness of atom distribution. At sufficiently high temperatures, atoms in an n-component alloy are assumed to be randomly distributed in the liquid state. According to Boltzmann’s equation, the configurational entropy of the alloy can be calculated as[3]:
where kB is the Boltzmann constant, w is the thermodynamic probability, R is the gas constant, and xi is the mole fraction of the ith element. Consequently, the alloy’s entropy increases with the number of constituent elements.
HEAs exhibit four critical characteristics: high entropy, lattice distortion, sluggish diffusion, and the cocktail effect[2], which enhance mechanical properties such as strength, ductility, and toughness, as well as electromagnetic properties such as superconductivity, shielding, and corrosion resistance[4].
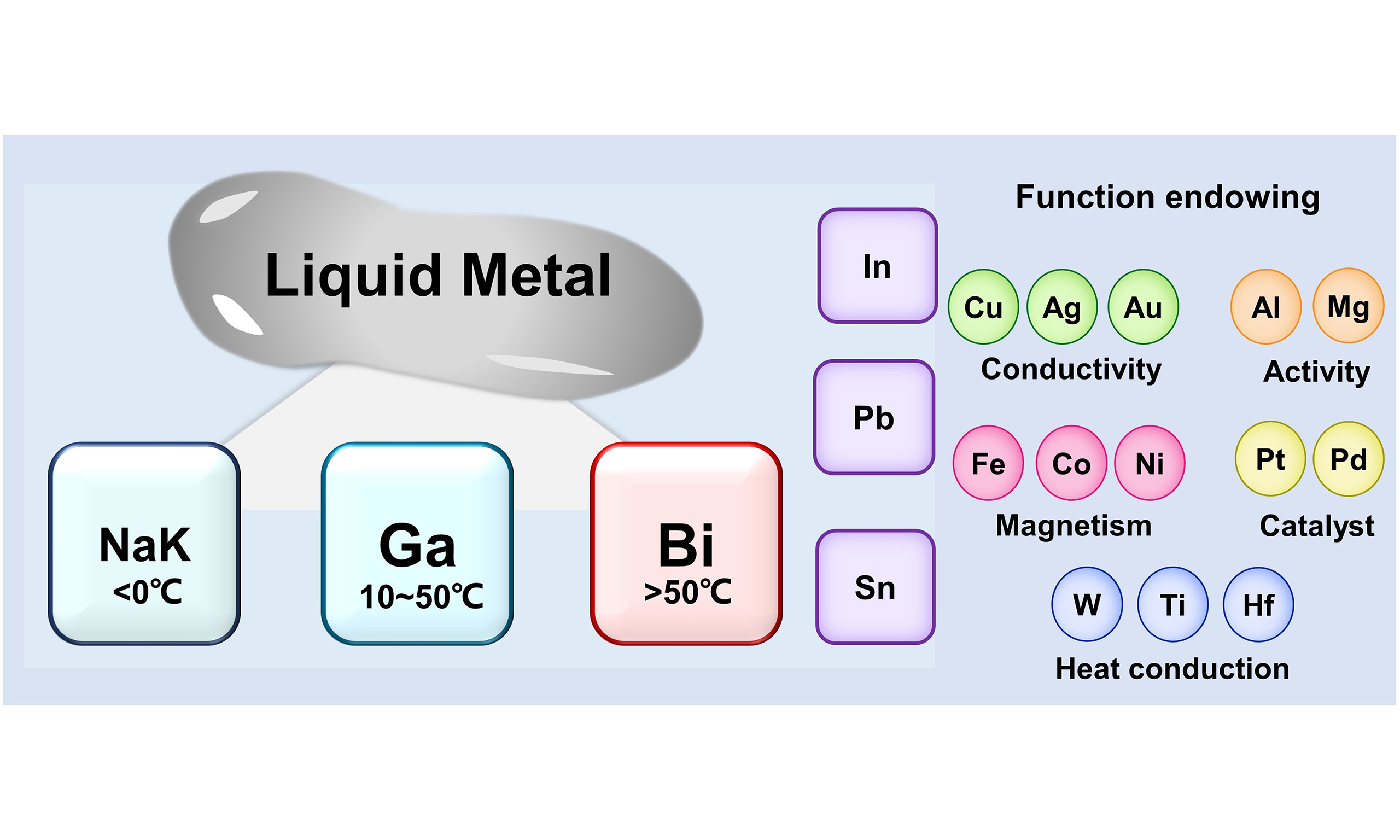
However, most elements used in HEAs have high melting points, making their preparation and application challenging. Conventional HEAs are typically prepared using high-temperature or energy-intensive methods such as arc melting, mechanical alloying, and physical vapor deposition. Although these approaches enable the fabrication of multicomponent alloys, they often require extreme conditions, limiting scalability and compositional flexibility. With the advancement of smart materials, there is a growing demand for alloys that are easy to synthesize, integrate, and apply. Room-temperature liquid metals (RTLMs), which melt below
CONCEPTS OF RTLMS
Binary and ternary RTLMs are most common due to their ease of fabrication, predominantly consisting of Ga-based alloys (melting point < 30 °C) and Bi-based alloys (> 40 °C). To expand the usable temperature window and tune material properties, quaternary and quinary systems incorporate additional elements such as Sn, Pb, Ca, or Cd. Na-K alloys have sub-zero melting points but are highly reactive, limiting their handling and practical applications.
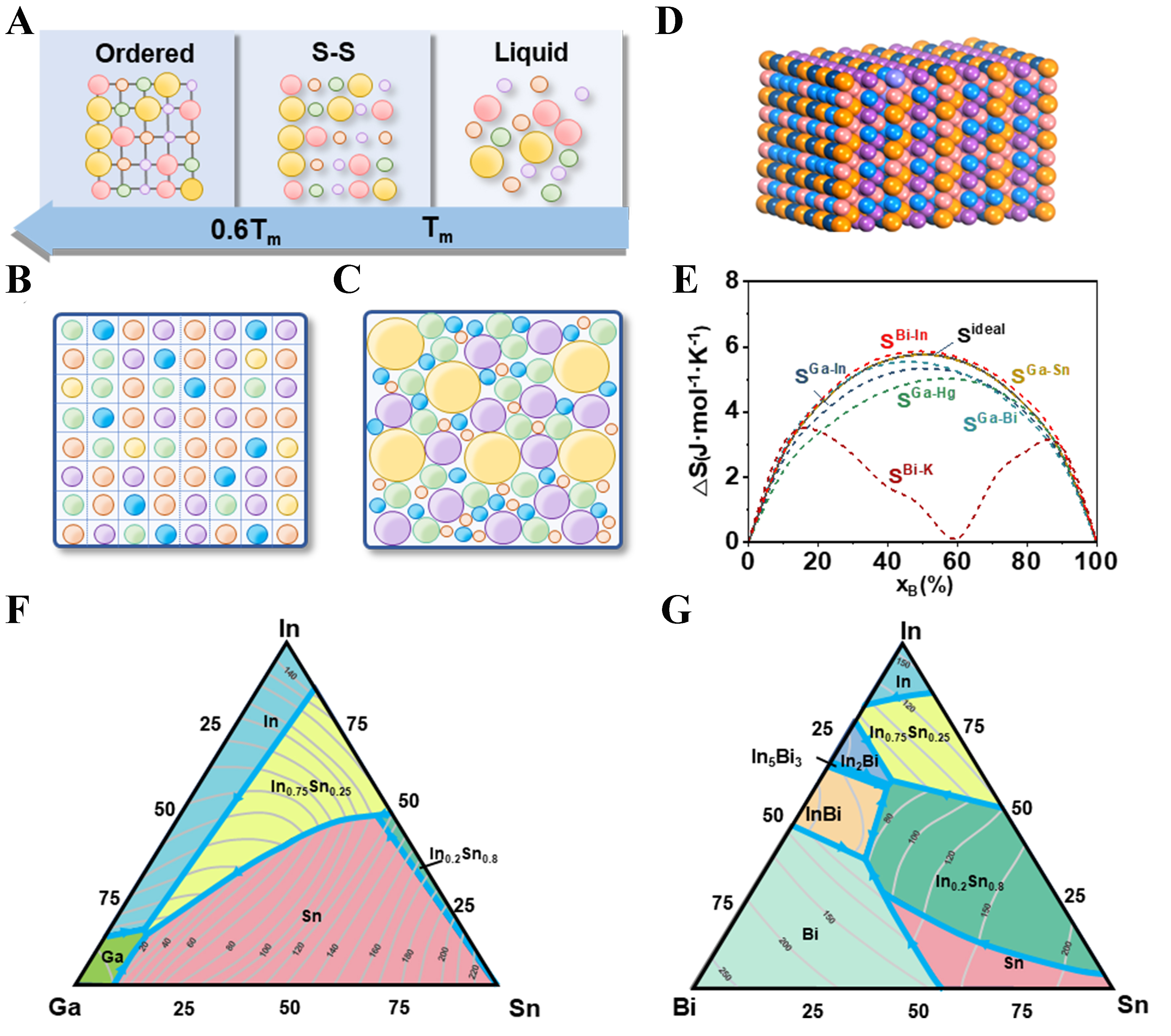
Owing to their low melting points, RTLMs can reach appreciable configurational entropy (∆S_conf) and are often grouped into low (< 1.0 R), medium (1.0-1.5 R), and high (> 1.5 R) entropy regimes. Their liquid state further enables the incorporation of additional elements, providing a practical medium for exploring multicomponent alloy chemistries. RTLMs also undergo phase transitions at relatively low temperatures, allowing solidification processes to be studied and controlled under mild conditions. As depicted in the phase evolution model for HEAs [Figure 1A], cooling induces a transformation from a liquid with local ordering to a solid solution, and eventually to a disordered multiphase solid due to segregation, ordering, and spinodal decomposition. In Ga-based systems, supercooling can maintain a liquid state far below the nominal melting point, facilitating elemental mixing and enabling direct observation of solidification behavior. In real multicomponent alloys, atomic-size mismatch leads to non-ideal packing and increased disorder relative to an ideal lattice model [Figure 1B and C][6]. Figure 1D shows a three-dimensional (3D) visualization of possible atomic arrangements in HEAs. Thus, when considering actual alloy systems, the excess entropy SE contributed by these components must also be accounted for. The total molar entropy ST can be expressed as[6]:
Figure 1. (A) Solidification process in a typical HEA, showing transition from liquid to solid solution and then to a multiphase disordered solid due to segregation and ordering. S-S represents the solid solution phase. Tm is the melting point; (B) Ideal atomic configuration with equal-sized atoms arranged in a regular lattice structure[6]; (C) Real atomic configuration in HEAs with varied atomic sizes, leading to random packing and increased entropy[6]; (D) 3D visualization of atom distribution in HEAs, showing disordered arrangements that contribute to excess entropy; (E) Comparison of phase behaviors in Ga-In and Bi-K systems, illustrating different entropy trends due to IMC formation. SA-B represents the total molar entropy ST of different binary systems; (F) Phase diagram of the Ga-In-Sn ternary alloy system with varying compositions and temperatures[7]; (G) Phase diagram of the Bi-In-Sn ternary alloy system[7]. (B and C) Adapted with permission from Ref.[6]. Copyright 2014 Elsevier. (F and G) Adapted with permission from Ref.[7]. Copyright 2001 Elsevier. HEA: High-entropy alloy; 3D: three-dimensional; IMC: intermetallic compound.
where SC represents the ideal configurational entropy. SE mainly arises from atomic size mismatch in packing, atomic vibrations, and magnetic or electric effects.
POTENTIAL OF LM-ENABLED METALLURGY
Previous studies have shown that Ga can form solutions with many metals such as Zn, Al, and Si, without producing IMCs, which aligns well with the design principles of HEAs. In contrast, Bi-based alloys tend to form IMCs with complex structures. Figure 1E shows some typical binary systems based on Ga and Bi, respectively. Additionally, the phase diagrams of the Ga-In-Sn [Figure 1F] and Bi-In-Sn [Figure 1G] ternary systems are presented, showing the compositions of alloys at different component ratios and temperatures[7].
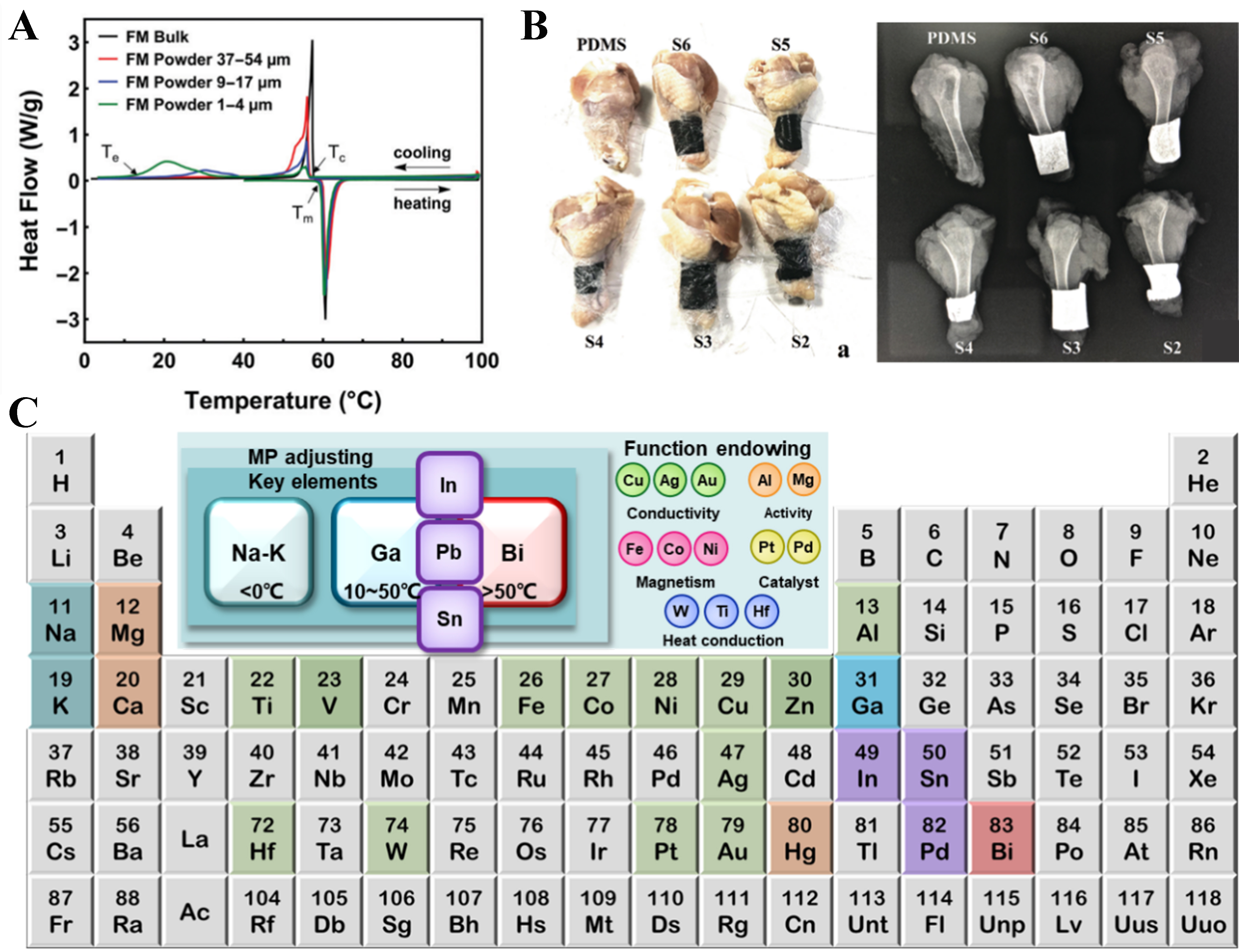
The quaternary BiInSnZn system has attracted attention for its phase-transition behavior and mechanical performance. Constructed from the Bi-In-Sn, Bi-In-Zn, Bi-Sn-Zn, and In-Sn-Zn subsystems, it shows relatively limited IMC formation compared with many multicomponent alloys, making it a promising candidate for further alloy development. Liu et al. used an equiatomic BiInSnZn alloy (melting point ~80 °C) as a low-melting, Pb-free solder and as a phase-transition component for stiffness-tunable materials[8]. Although supercooling can hinder precise triggering on phase transition, Zn incorporation suppresses supercooling, reduces thermal hysteresis, and improves suitability for stiffness regulation. The size-dependent thermal behavior of this alloy is summarized in Figure 2A[9].
Figure 2. (A) Thermal performance of BiInSnZn alloys with varying particle sizes, demonstrating size-dependent regulation of thermal behavior[9]. Tc and Tm represent the freezing onset temperature and melting onset temperature, respectively; (B) X-ray imaging effect of GaInSnBiPb alloys with different compositions, illustrating the combination of high radiation shielding capability and elasticity[13]; (C) LM-enabled metallurgy: design principles and performance potential. By combining these alloys, it is possible to produce new alloys with desired properties, potentially for use in the production of medical radioactive sources. (A) Adapted from Ref.[9]. Licensed under CC BY-NC 3.0. (B) Adapted with permission from Ref.[13]. Copyright 2021 Wiley-VCH GmbH. LM: Liquid metal; PDMS: polydimethylsiloxane; MP: multiprincipal.
Driven by the increasing demand for lead-free solders, BiInSn is often used as a base alloy that can be alloyed with additional elements to achieve functional tunability. For example, Ag is commonly incorporated due to its chemical stability and high thermal/electrical conductivity. A non-equilibrium model has been established for the BiInSnAg quaternary system to predict and analyze its melting behavior, providing a pathway for the rational design of low-melting, lead-free solders. In comparison, GaInSnZn exhibits a lower melting point than BiInSnZn, likely due to the solubility of Zn in Ga (~3.9 at%), enabling straightforward melting point regulation via Zn content. Benefiting from favorable wettability on Ti3C2Tx MXene, GaInSnZn can be stably printed to form flexible lithium-ion battery anodes. Similar designs using siloxane-based matrices have also been explored to enhance conductivity and self-healing, thereby extending liquid metal applications toward low-temperature and low-cost energy devices[10,11].
Beyond directly melting and mixing pure metals, as in conventional metallurgy, the relatively low melting points of liquid metals enable liquid-liquid alloying, in which two molten alloys are blended in the liquid state to generate a new alloy system. Yu et al. developed a Bi26In61Sn9Ga4 alloy with a melting point of 52 °C for use as a 3D printing ink[12], enabling rapid metallic printing near room temperature with higher mechanical performance than cast counterparts (~17.81 vs. ~15.78 GPa)[12]. Increasing Bi content drives a transition from amorphous to crystalline phases, which affects wettability and adhesion and supports the design of deformable GaBiInSn-based flexible electrodes. In parallel, Pb-alloyed liquid metals have been investigated for radiation-shielding coatings, achieving higher mass attenuation than pure Pb. When combined with polydimethylsiloxane (PDMS), the resulting composites provide both enhanced shielding and favorable elasticity, as demonstrated by the X-ray imaging results in Figure 2B[13].
Recently, liquid metals have also been employed as reaction media for the synthesis of HEA functional nanoparticles. Cao et al. proposed a strategy using liquid Ga to fabricate HEA nanoparticles with high element inclusiveness under relatively mild conditions (~923 K)[14]. The HEA particle size can be tuned by the Ga nanoparticle template, enabling controlled synthesis. More broadly, liquid metals can act as nanoreactors and catalytic media at the atomic or molecular scale, offering a versatile approach to catalyst design.
OUTLOOK
In this article, we propose LM-enabled metallurgy based on RTLMs as a practical route to access HEA-relevant compositional spaces under reduced thermal budgets, and summarize a design framework spanning element selection, fabrication routes, and functional evaluation [Figure 2C]. Several limitations must be addressed before broader translation. Element immiscibility can restrict the compositional space and induce phase separation or unstable microstructures. Ga- and Bi-based systems are susceptible to oxidation, which compromises stability and complicates processing. In addition, alloys obtained through reduced-temperature routes may exhibit limited high-temperature robustness compared with those produced via conventional high-temperature metallurgy, thereby restricting their use in demanding structural environments. These issues motivate strategies such as encapsulation or surface passivation to mitigate oxidation, composition tuning to improve miscibility, and hybrid design approaches to enhance thermal and chemical stability.
New strategies for metallurgy
From a metallurgical standpoint, the key value of LM-enabled routes lies in using liquid metals as intermediate media with intrinsic fluidity, surface activity, and wettability. RTLMs can facilitate multielement dissolution and mass transport, reduce kinetic barriers, and accelerate homogenization at reduced temperatures. They can also mediate interfacial reactions that promote the incorporation of otherwise immiscible elements, thereby expanding accessible compositional combinations[15]. Meanwhile, the interfacial reaction mechanisms at liquid metal-solid boundaries remain insufficiently understood, and their clarification is essential for achieving controllable synthesis and reproducible phase outcomes. Current studies are still concentrated on a few RTLM families (e.g., Ga-In, Ga-In-Sn, Bi-In-Sn), and generalizable composition-phase-property relationships remain lacking. Establishing a materials genome that links composition, phase evolution during liquid-to-solid transitions, interfacial chemistry, and functional performance across alloy families will therefore be a critical next step.
AI-assisted calculation methods and theoretical models
Current work on LM-enabled metallurgy is still fragmented, largely confined to a few RTLM families such as Ga-In, Ga-In-Sn, and Bi-In-Sn. Systematic mapping of composition, melting behavior, stability, and functional performance remains limited. Building a comprehensive materials genome that links composition, phase evolution, interfacial chemistry, and properties across alloy families would provide a foundation for rational design. In parallel, artificial intelligence (AI) offers new opportunities; however, modeling approaches must extend beyond conventional solid-state HEA frameworks to account for liquid-state transport and liquid-to-solid transitions. Coupling machine learning with thermodynamic and kinetic modeling could accelerate composition screening, phase prediction, and experimental design with improved efficiency[16].
Potential applications
Although LM-enabled metallurgy is still at an early stage, its combination of low-melting fluidity and multicomponent design offers broad prospects. Fluidity of the liquid metal enables large deformability, reconfigurability, and self-healing, while multielement chemistry provides enhanced stability, strength, and functional tunability. These features support applications such as adaptive actuators and stretchable conductors for soft robotics, printable inks for additive manufacturing, and reconfigurable or self-healing components for smart materials. They also enable conformal interfaces for biomedical devices and bioelectronics. Moreover, their multielement electronic structures and liquid-state dynamics open new opportunities in energy storage, thermal management, and catalysis. At present, however, most reported systems remain relatively simple ternary or quaternary Ga-, In-, or Sn-based alloys. Expanding toward more complex and function-driven compositions - guided by systematic materials genome mapping and AI-assisted prediction - will be essential for practical translation.
DECLARATIONS
Acknowledgments
The authors thank Prof. Jing Liu for his insightful contributions to the conceptual design during discussions.
Authors’ contributions
Conceptualization: Yuan, B.
Supervision: Yuan, B.; Wang, H.
Writing - original draft: Yuan, B.; Liu, C.
Writing - review and editing: Yuan, B.; Tang, S.; Wang, H.
Availability of data and materials
Not applicable.
AI and AI-assisted tools statement
During the preparation of this manuscript, the AI tool ChatGPT (version GPT-5, released 2025-08-07) was used solely for language editing. The tool did not influence the study design, data collection, analysis, interpretation, or the scientific content of the work. All authors take full responsibility for the accuracy, integrity, and final content of the manuscript.
Financial support and sponsorship
This work was supported by the National Natural Science Foundation of China (Grant No. 52203360); Basic Scientific Research Operating Funds Project of Beihang University (Grant Nos. JKF-20240588, KG16280101, and KG16388101); and the science and technology cooperation funding between Nanshan District (Shenzhen) and Tsinghua SIGS.
Conflicts of interest
All authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
REFERENCES
1. Lei, Z.; Liu, X.; Wu, Y.; et al. Enhanced strength and ductility in a high-entropy alloy via ordered oxygen complexes. Nature 2018, 563, 546-50.
2. Hsu, W. L.; Tsai, C. W.; Yeh, A. C.; Yeh, J. W. Clarifying the four core effects of high-entropy materials. Nat. Rev. Chem. 2024, 8, 471-85.
3. Ren, J. T.; Chen, L.; Wang, H. Y.; Yuan, Z. Y. High-entropy alloys in electrocatalysis: from fundamentals to applications. Chem. Soc. Rev. 2023, 52, 8319-73.
4. Li, Z.; Pradeep, K. G.; Deng, Y.; Raabe, D.; Tasan, C. C. Metastable high-entropy dual-phase alloys overcome the strength-ductility trade-off. Nature 2016, 534, 227-30.
5. Shen, Q.; Jiang, M.; Wang, R.; et al. Liquid metal-based soft, hermetic, and wireless-communicable seals for stretchable systems. Science 2023, 379, 488-93.
6. Ye, Y. F.; Wang, Q.; Lu, J.; Liu, C. T.; Yang, Y. The generalized thermodynamic rule for phase selection in multicomponent alloys. Intermetallics 2015, 59, 75-80.
7. Hoch, M. Thermodynamic behavior of inorganic and organic systems with eight and more components. Calphad 2001, 25, 253-66.
8. Liu J, Yi L. Liquid metal biomaterials: principles and applications, 1st ed.; Springer Singapore, 2018.
9. Ma, J.; Zhang, P. Supercooling suppression of phase change liquid metal–polydimethylsiloxane soft composites. Mater. Adv. 2021, 2, 7437-44.
10. Wei, C.; Fei, H.; Tian, Y.; et al. Room-temperature liquid metal confined in MXene paper as a flexible, freestanding, and binder-free anode for next-generation lithium-ion batteries. Small 2019, 15, e1903214.
11. Bai, J.; Jin, X.; Yang, H.; Qiao, J. A low melting high entropy alloy with conformal electroconductivity for flexible electronic circuits. J. Alloys. Compd. 2022, 919, 165736.
12. Yu, Y.; Liu, F.; Liu, J. Direct 3D printing of low melting point alloy via adhesion mechanism. Rapid. Prototyp. J. 2017, 23, 642-50.
13. Wang, K.; Hu, J.; Chen, T.; et al. Flexible low‐melting point radiation shielding materials: soft elastomers with GaInSnPbBi high‐entropy alloy inclusions. Macromol. Mater. Eng. 2021, 306, 2100457.
14. Cao, G.; Liang, J.; Guo, Z.; et al. Liquid metal for high-entropy alloy nanoparticles synthesis. Nature 2023, 619, 73-7.
15. Liang, J.; Chen, S.; Ni, E.; et al. High-entropy alloy array via liquid metal nanoreactor. Adv. Mater. 2024, 36, e2403865.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data


















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.