Bioinspired mechanosensitive ion channels: design principles and emerging applications
Abstract
Mechanosensitive ion channels (MSCs) enable living systems to convert mechanical forces into electrochemical signals, thereby providing fundamental support for essential physiological processes such as touch, hearing, osmoregulation, and blood-pressure sensing. Recent advances in cryo-electron microscopy and single-molecule biophysics have revealed the structural architectures and force-gating mechanisms of diverse MSC families, including mechanosensitive channel of large conductance (MscL), Piezo, transient receptor potential (TRP), two-pore domain potassium (K2P), and osmosensitive calcium-permeable/transmembrane 63 (OSCA/TMEM63). Inspired by these natural MSCs, bioinspired MSCs (BMSCs) have emerged as a rapidly growing kind of artificial systems capable of translating membrane tension, pressure, or deformation into controllable ion transport. In this Review, we outline the structural features and mechanotransduction mechanisms of representative natural MSCs. We systematically discuss current construction strategies for BMSCs, spanning (i) electrokinetic modulation; (ii) pore/channel deformation, and (iii) molecular conformational gating. Finally, we introduce emerging applications of these systems in smart drug delivery, ultrasensitive mechanical sensing, and memristor devices. By bridging molecular mechanotransduction with synthetic nanofluidic and functional materials, BMSCs provide a versatile platform for mechanically adaptive ion transport and open new opportunities for next-generation biomaterials and intelligent electronic systems.
Keywords
INTRODUCTION
Mechanosensitive ion channels (MSCs) are a type of transmembrane protein capable of converting mechanical stimuli (such as membrane tension, pressure, or shear stress) into electrochemical signals. They play essential roles in enabling cells to sense and respond to their mechanical environment[1]. These channels are ubiquitously present in both prokaryotic and eukaryotic organisms and are involved in a broad range of physiological processes, including touch and hearing perception, osmoregulation, cell migration, cardiovascular regulation, and nociception[2,3]. In recent years, with advances in cryo-electron microscopy and single-molecule biophysical techniques, researchers have been able to reveal their structures, gating mechanisms, and physiological functions. Representative examples include the mechanosensitive channel of large conductance (MscL)[4], the Piezo channels[5], transient receptor potential (TRP) channels[6], two-pore domain potassium (K2P) channels[7], and osmosensitive calcium-permeable/transmembrane 63 (OSCA/TMEM63) channels[8,9]. In nature, MSCs respond to membrane tension or changes in the membrane environment by undergoing conformational rearrangements[10]. These structural changes allow precise control of ion permeability. This efficient and rapid mechano-electrical conversion mechanism has inspired the design of intelligent materials and bioelectronic systems.
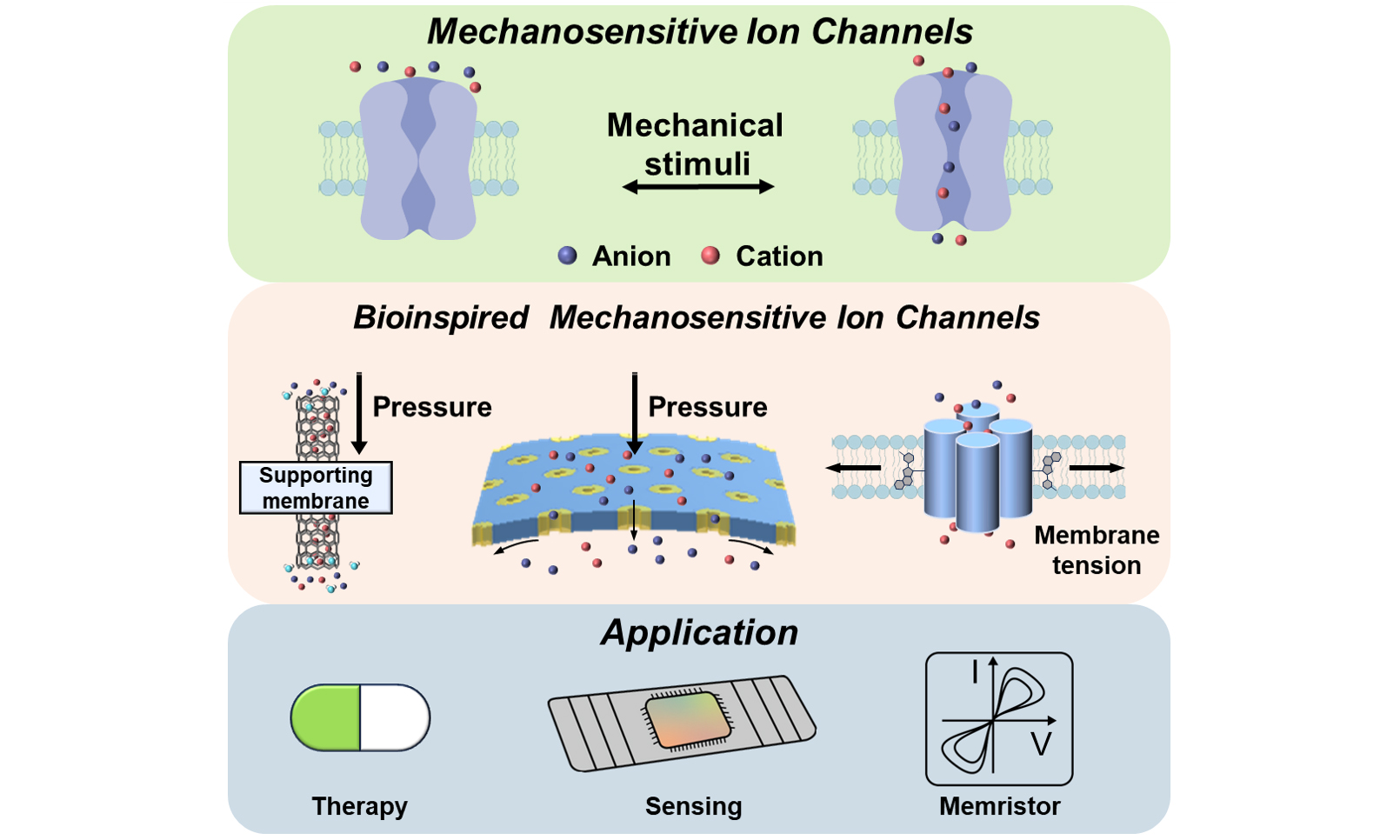
By studying the structures and mechanical regulation mechanisms of natural ion channels and extracting bioinspired principles from them, a foundation is established for the rational design of bioinspired MSCs (BMSCs). Researchers have begun to construct artificial systems that can regulate ion transport[11-16], detect mechanical signals[17], and interface with living tissues[18,19]. Such bioinspired systems hold great promise for applications in disease treatment, mechanical sensors, and memristor devices [Figure 1]. This review provides a comprehensive overview of the current development of BMSCs. We introduce the structural and functional features of natural MSCs and outline current design strategies for BMSCs, and discuss their emerging applications.
Figure 1. The design strategy of bioinspired mechanosensitive ion channels (BMSCs) and their applications. The green part contains the structures and response capabilities of the five family channels. The orange part contains the current strategies for constructing BMSCs. The electrokinetic modulators are mostly composed of glass, graphene, and carbon nanotubes. Reproduced with permission[11]. Copyright 2011 American Chemical Society. Pore/Channel deformation is typically constructed by exploiting SiN film or block-copolymer self-assembly to form well-defined nanochannels, and it responds to mechanical stimuli by changing the effective pore size. Reproduced with permission[15]. Copyright 2020, American Chemical Society. The molecular conformational gating nanochannels are mainly composed of amphiphilic molecules, butterfly-shaped molecules, six-helix-bundle DNA and pillar-shaped molecules, and mainly respond to membrane tension. Reproduced with permission[19]. Copyright 2025 Wiley-VCH. The blue part contains some applications of BMSC, including disease treatment, mechanical force sensors and nanofluidic iontronic memristors. MscL: Mechanosensitive channel of large conductance; TRPN: transient receptor potential channel, subfamily N; K2P: two-pore domain potassium; OSCA: osmosensitive calcium-permeable.
STRUCTURES AND MECHANISMS OF NATURAL MECHANOSENSITIVE ION CHANNELS
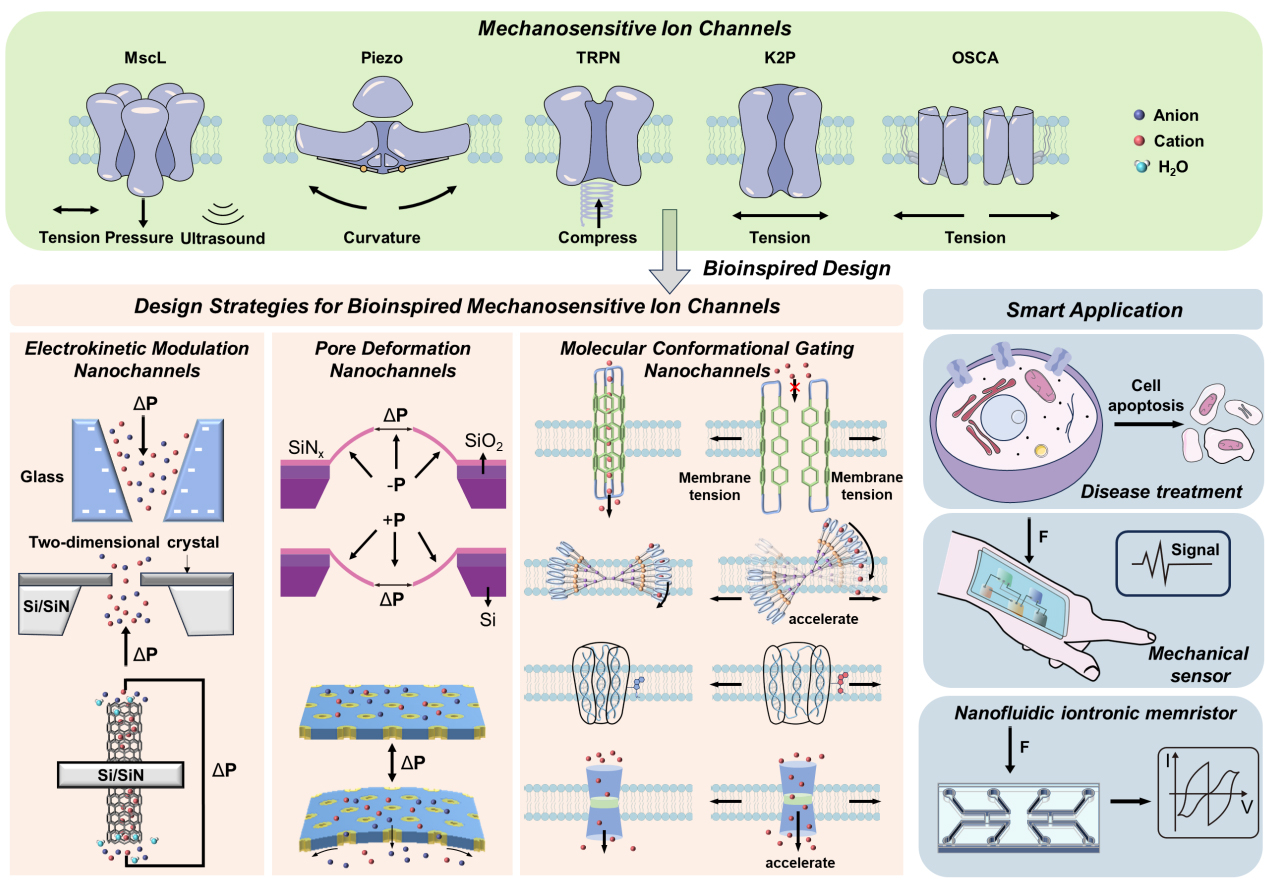
MSCs comprise a diverse array of membrane proteins that convert mechanical disturbances of the lipid bilayer into changes in ion permeability[3]. Different families use unique structural designs and gating mechanisms, yet they all achieve the same functional goal: transforming mechanical force into ionic current. Since there have been numerous comprehensive reviews[3,20,21] that have summarized the differences among various types of MSCs, we will merely provide a brief introduction and comparison of five representative families - MscL, Piezo channels, TRP channels, K2P family (e.g., TWIK-related K+ channel, TREK/TWIK-related arachidonic acid-activated K+ channel, TRAAK), and the OSCA/TMEM63 family - with respect to overall architecture, mechanogating mechanism, ion selectivity, and representative physiological roles.
Bacterial mechanosensitive channels: MscL and MscS
In bacterial cells, MSCs provide an essential relief mechanism during rapid osmotic shock[22]. The large-conductance mechanosensitive channel (MscL) and the small-conductance mechanosensitive channel (MscS) from Escherichia coli were among the first channels shown to open in response to increased membrane tension and to act as cellular “safety valves”[4,22].
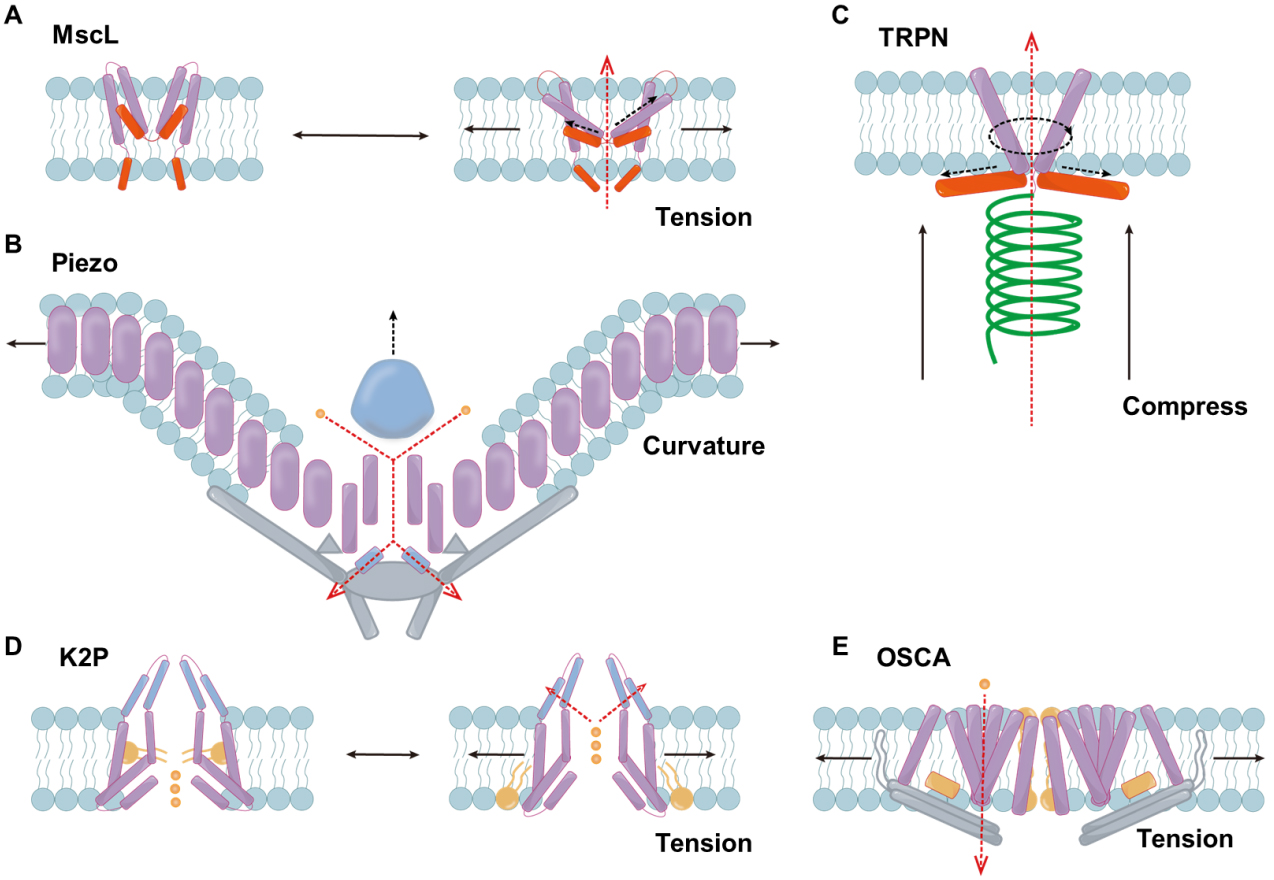
MscL is a pentameric, large-conductance channel that opens in response to increased membrane tension during osmotic shock. Each MscL subunit contains two transmembrane α-helical domains (TM1 and TM2) connected by an extracellular loop. The five TM1 helices together form the pore of the ion channel, while TM2 interacts with the lipid bilayer[23,24]. When external osmotic pressure decreases sharply, TM2 senses changes in membrane tension and transmits the signal to TM1, which rotates clockwise by about 100° and inserts into the bilayer (Figure 2A, TM1 in purple). The pore then expands significantly (to approximately 30 Å), allowing cytoplasmic solutes and ions to escape, thereby reducing intracellular osmotic pressure and preventing cell rupture[4,25].
Figure 2. Illustration of the structure of mechanosensitive ion channels from different families. (A) Schematic diagram of the response mechanism of MscL; (B) The structure of the Piezo channel; (C) The structure of the TRPN channel; (D) The response mechanism of K2P; (E) The structure of the OSCA channel. Black solid line: direction of mechanical force. Dark blue: domain. Blue: membrane lipids. Purple: TM helices. Orange: lipid acyl. Red: amphipathic helices. Gray: bundled domain. Green: tether structure. Gold: ions. Black dotted line: change in protein domain. Red dotted line: direction of ion transport. MscL: Mechanosensitive channel of large conductance; TRPN: transient receptor potential channel, subfamily N; K2P: two-pore domain potassium; OSCA: osmosensitive calcium-permeable; ΔP: pressure difference.
In addition to MscL, bacteria also employ MscS for osmotic regulation[4]. MscS is different in structure from MscL. Each MscS subunit contains three transmembrane (TM1, TM2, and TM3) helices, and seven subunits assemble into a homoheptamer[26,27]. The peripheral helices TM1 and TM2 are positioned on the outer side of the channel and arranged in an antiparallel manner with a crossing angle of approximately 165°, together forming the sensor paddle structure. TM3 constitutes the pore-lining helix of the channel and can be divided into two segments, TM3a and TM3b. TM3a forms the inner wall of the pore, whereas TM3b connects to the cytoplasmic domain[28,29]. Recent MscS structures resolved in lipid nanodiscs reveal lipid-filled pockets surrounding the sensor paddle and pore-forming domains, and these bound lipids appear to be important for gating under mechanical stimuli[30,31]. As membrane tension increases, these pockets become compressed and the associated lipids are displaced, allowing the transmembrane helices to tilt and flatten, thereby expanding the pore[27].
Propeller-shaped Piezo channels
The discovery of the Piezo1 and Piezo2 channels marked a major leap in understanding mechanotransduction in eukaryotes, particularly in animals[5]. Piezo channels mediate diverse eukaryotic mechanosensory functions - Piezo2 is essential for touch and proprioception, while Piezo1 functions in endothelial shear sensing, red blood cell volume regulation, and other physiological processes[32,33].
These channels are large (~ 2,500 residues), assemble as homotrimers, and adopt a distinctive three-bladed, propeller-shaped architecture, each “blade” embedded in the lipid bilayer and contributing to membrane curvature[34,35]. High-resolution cryo-electron microscopy structures revealed Piezo1 as a three-bladed propeller or “triskelion” with a central ion-conducting pore and peripheral curved transmembrane “blades” that deform the membrane into a nano-dome [Figure 2B][36,37]. Structural features such as the peripheral dome, anchor, beam, and cap modules have been identified as mechanotransduction elements[34,36,38,39]. Lipid composition, membrane stiffness, and cytoskeletal interactions all modulate gating behavior. Piezo2 shares a similar overall architecture but with distinct extracellular loops and intracellular elements that tune its activation and inactivation kinetics, correlating with its specialized role in touch and proprioception[39-41].
Piezo channels are nonselective cation channels[42]. The changes in membrane tension or curvature alter the relative arrangement of the blades and the central pore, producing a lever-like gating mechanism that opens a nonselective cation pore permeable to alkali ions (Li+, Na+, K+, Cs+) and divalent (Mg2+, Ca2+, Ba2+)[36,39,42-44]. It is rapidly activated upon sensing changes in lipid membrane curvature or tension, and then rapidly inactivated, thus allowing cells to encode dynamic mechanical cues[5,45,46]. These properties provide new design principles for the development of bioinspired mechanosensors with rapid and transient ionic response capabilities.
TRP channels: diverse sensors with both direct and indirect mechanosensitive roles
The TRP channel superfamily encompasses many members that detect various stimuli, including thermal, chemical, and mechanical cues[3,47-49]. The typical structural organization of TRP channels is that of a tetramer assembled from identical or homologous subunits. Each subunit contains six transmembrane helices, and both the N- and C-terminal regions are located on the cytoplasmic side of the membrane. The TM5 and TM6 helices are joined by a short extracellular loop, which together constitute the cation-conducting pore[47].
Several TRP subtypes (e.g., TRPA1[50,51], TRPV4[52], and TRPN[53,54] in various organisms) have been implicated in mechanotransduction either through direct mechanical activation or via second-messenger pathways downstream of non-channel mechanical sensors. For instance, TRPV4 integrates osmotic and shear stress signals, but its mechanosensitivity often requires intermediary pathways such as integrin activation or phospholipase-dependent lipid signaling[52]. Conversely, recent studies have shown that human TRPA1 (hTRPA1) can be directly gated by membrane stretch when reconstituted into defined lipid bilayers[50]. TRP channels are typically activated indirectly by mechanical stimuli, whereas TRPN represents one of the few TRP subgroups that can be directly gated by mechanical forces owing to a well-defined force-transmission architecture - for example, an ankyrin repeat-rich cytoplasmic domain mechanically coupled to the pore module (Figure 2C, tether structure in green)[54]. The force on the TRPN channel is transmitted through the tether to the extracellular matrix, the cytoskeleton, or both[54].
TRP channels thus represent “hybrid” mechanosensors whose mechanical response is strongly context-dependent. Their mechanosensitivity is heterogeneous across family members and often modulated by lipids, cytoskeletal attachments, or intracellular signaling. For many TRPs, distinguishing direct force-gating from indirect mechanosensitive modulation remains a challenge[52]. This type of multimodal gated channel that responds to multiple environmental signals provides more design ideas for artificial ion channels.
K2P channels (TREK/TRAAK): lipid-bilayer mediated mechanosensitivity
TREK-1, TREK-2, and TRAAK are mechanosensitive members of the two-pore-domain potassium (K2P) channel family that mediate background K+ currents, thereby stabilizing the membrane potential and reducing cellular excitability[55-57]. K2P channels are dimers of subunits with four transmembrane helices and two pore-loops each, forming an intrinsically heterotetrameric K+ selectivity ion channel capped by an extracellular helical dome that bifurcates the ion entry pathway[58,59]. The crystal structures of TRAAK and TREK reveal the existence of two distinct “up” and “down” conformations. In these conformations, the intracellular end of the TM helix (M4) swings relative to the lipid bilayer, while the hydrophobic pore openings open or close, and these pores can be filled with lipid acyl chains (Figure 2D, lipid acyl in red)[60,61]. Increased membrane tension or changes in lipid composition (such as different phospholipid acyl chains or head groups) cause the transmembrane helices to shift. This motion alters the protein cross-sectional area in the inner leaflet, so that increased membrane tension biases the ensemble toward the expanded “up” conformation[62]. These conformational changes allow K+ ions to flow outward.
The mechanogating mechanisms of K2P channels are generally classified into two categories. In the first, which is analogous to that of MscL, increased membrane tension shifts the channel toward conformations with a larger in-plane cross-sectional area. However, to preserve K+ selectivity, this change in cross-sectional area occurs predominantly in the portion of the channel located below the selectivity filter[62,63]. The structures of TREK-2 and TRAAK show that in the “down” conformations, the lipid tail chains can enter and partially block the pore cavity. After activation, it transforms into the “up” conformation, the lateral pores close, and the lipid cannot continue to occupy the pore cavity, resulting in enhanced K+ conduction[55,57,61,64,65].
Introducing hydrophobic pockets that can be reversibly occupied into nanopores or channel walls represents an attractive design strategy. Hydrophobic polymer chains or functionalized lipids can serve as steric plugs, and mechanical stretching can then modulate the volume or aperture of these pockets to achieve controllable opening and closing of the channel.
OSCA/TMEM63 family
OSCA and TMEM63 channels form a conserved family of eukaryotic mechanosensitive ion channels that convert high-threshold membrane tension and osmotic stress into Ca2+-dominated signals[9,66]. Plant OSCA proteins are key components of hyperosmotic and mechanical stress responses[67], whereas mammalian TMEM63A-C are associated with the breath[68], hearing[69], and myelination defects in infants[70].
The cryo-electron microscopy structure reveals that OSCA1.2 forms a trapezoidal homodimer, with each subunit containing 11 transmembrane helices, assembling into a central cavity and lateral channels that can be accessed by lipids (Figure 2E, inter-subunit gaps in red, a two-polar helix in purple, a bundled domain in gray)[3,71-77]. In contrast, the TMEM63 protein in mammals is a narrow ion channel formed by a single unit, and it has a higher sensitivity to pressure[9,66]. Recent studies have shown that the activation mechanism of the OSCA/TMEM63 channel is the “force-from-lipids” mechanism[74,76,78]. Under membrane tension, the pore and interfacial lipids are pushed outward, forming a mixed protein-lipid pore, while the extracellular domain expands in a manner resembling a blooming flower[74,76]. In the resting state, the interface lipids and the cytoplasmic lipids re-enter the channel, forming a tightly closed pore with extremely low electrical conductivity and a high activation threshold[74,76]. By introducing tension-sensing amphiphilic helices and mechanically-driven gating elements in the synthetic channel, it is expected to simulate the threshold characteristics and robustness of the OSCA/TMEM63 type, laying the foundation for the development of bioinspired nanofluidic devices and mechanically activated release systems.
Although different families vary in terms of oligomeric state, topological structure, and ionic selectivity [Table 1 and Figure 2], their common essence is to convert mechanical disturbances such as membrane tension/curvature/osmotic pressure into changes in permeability and transient ionic currents through force-conducting structural units. An in-depth study of the structure and gating mechanism of natural MSCs can provide more inspiration for the development of BMSCs.
Comparative features of representative mechanosensitive channel families
| Family | Oligomeric state/topology | Gate trigger | Ion selectivity | Representative physiological roles | Ref |
| MscL | Pentamer; simple TM helix bundle. | Direct membrane tension | Nonselective | Microbial osmotic shock protection | [4] |
| Piezo1/2 | Homotrimer; propeller-shaped | Membrane curvature/tension | Nonselective cation | Touch, proprioception, shear sensing | [5] |
| TRP | Tetrameric (typical) | Direct force or indirect biochemical pathways. | Nonselective cation | Diverse sensory roles (touch, hearing) | [79] |
| K2P | Dimer of subunits with two pore domains each | Lipid-mediated membrane tension | K+ selective | Neuronal excitability, pain modulation. | [80] |
| OSCA | Dimeric assemblies | Osmotic/ Lipid-mediated membrane tension | Nonselective cation | Plant osmosensing | [76] |
| TMEM63 | Monomeric | Lipid-mediated membrane tension | Nonselective cation | Mammalian respiration control, hearing | [9,66] |
DESIGN STRATEGIES FOR BIOINSPIRED MECHANOSENSITIVE ION CHANNELS
The term “bioinspired” is intended to address problems in biological and chemical research by using elements of the structural and/or functional aspects of biological systems. The term "biomimetic" is intended to replicate, as accurately as possible, the specific parts of a biological system in terms of structure and/or function[81]. In a conservative sense, many artificial platforms are functionally analogous to biological mechanosensitive channels at the molecular/nanoscale, rather than strictly “biomimetic” in reproducing protein-like conformational gating pathways[81,82]. Therefore, we use BMSCs as the general term for such artificial ion channels with mechanosensitivity. Bioinspired ion channels are gradually achieving intelligent ion transport behaviors similar to those of natural channels, including high selectivity, gating, and rectification mechanisms[83]. BMSCs aim to convert mechanical inputs acting on a membrane or device (e.g., an applied pressure difference ΔP, bending/curvature, or membrane tension) into electrochemical or optical outputs under a defined electrical bias. Based on the response mechanism of the channel, the current strategies can be roughly classified into the following three categories: (i) electrokinetic modulation; (ii) pore/channel deformation; and (iii) molecular conformational gating.
Electrokinetic modulation
Nanochannels exhibiting electrokinetic modulation show mechanosensitive-like ionic responses primarily through flow-charge coupling and interfacial electrostatics, rather than through a discrete open/closed gating transition. In charged nanopores and nanoslits, a pressure difference (ΔP) across the channel drives liquid flow that advects counterions within the electrical double layer, generating a streaming current or potential and continuously tuning conductance, selectivity, and rectification[84-86].
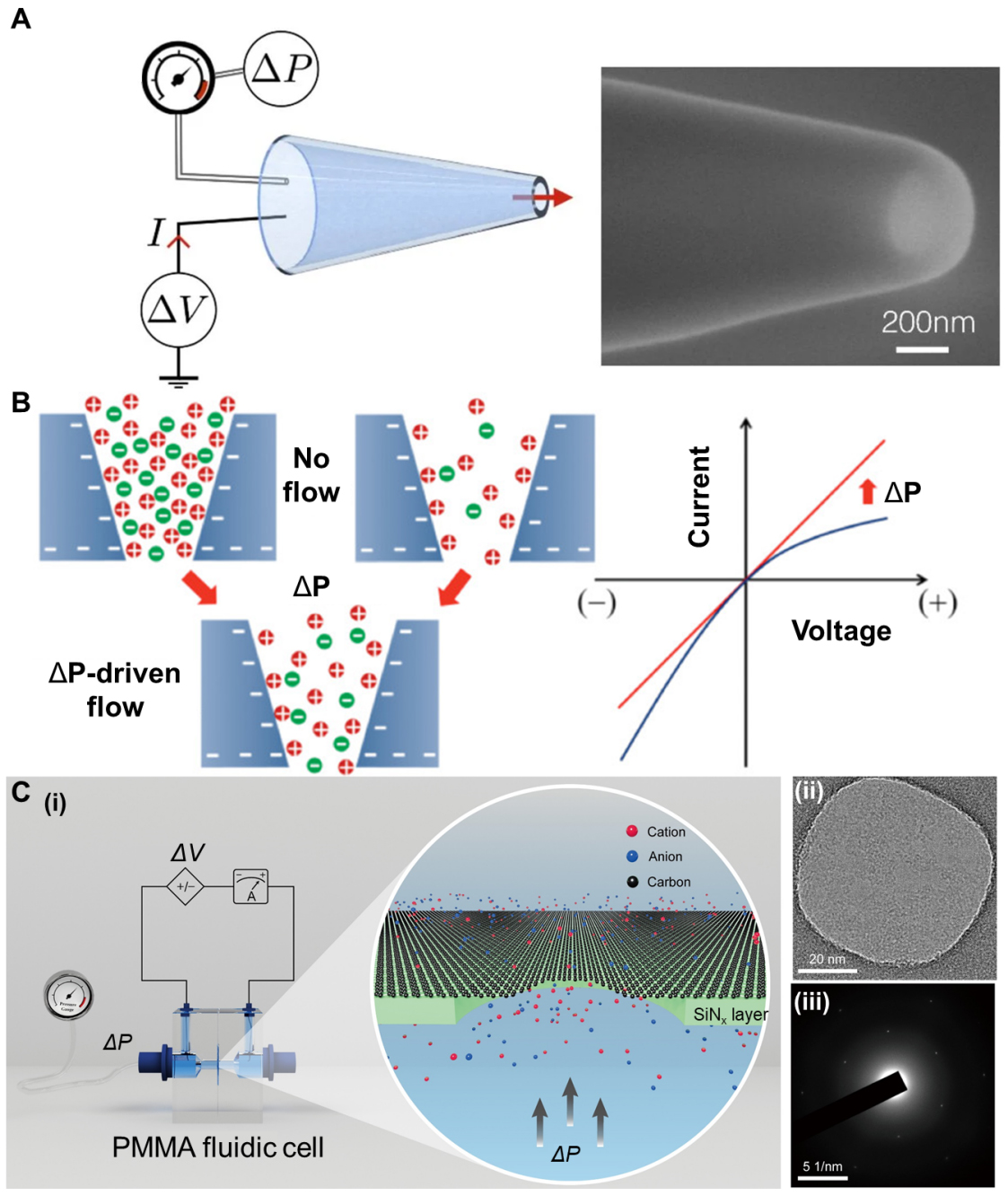
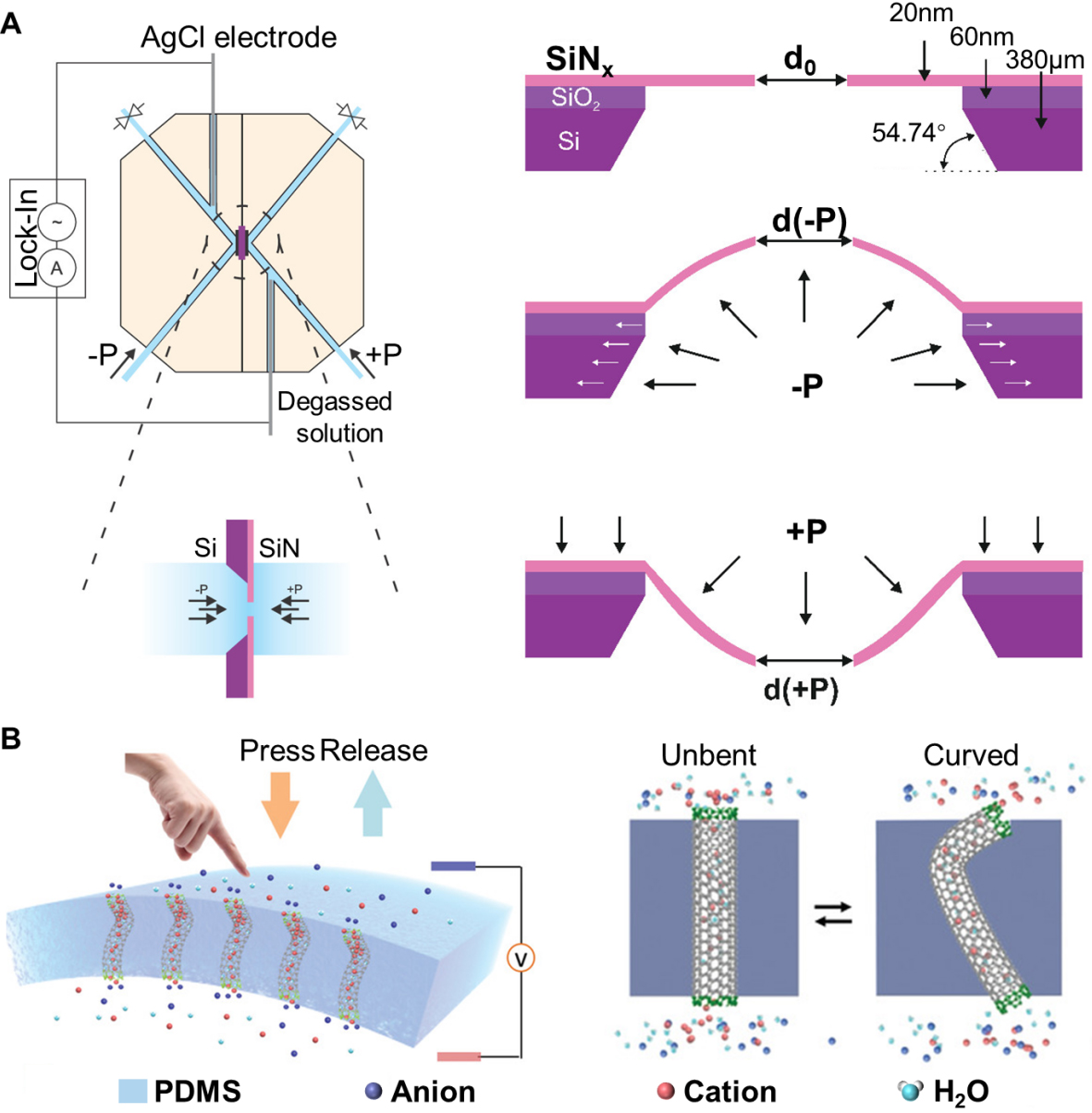
Glass nanochannels. Glass nanopores (typically pulled nanopipettes or etched conical pores in silica, Figure 3A) combine a mechanically rigid scaffold with an inner surface dominated by silanol groups[85,87,88]. Applying a static fluid pressure gradient to the charged silica nanochannels generates a measurable streaming current and potential, effectively converting mechanical work into electricity[84]. The key mechanism lies in the diffusion movement of counterions within the electric double layer under the pressure effect. This has similar properties to the force control mechanism observed in biological cells derived from lipids. Lan et al. investigated conical glass nanopores [Figure 3B] and revealed that ion current rectification (ICR) - the diode-like difference between forward and reverse currents - changes systematically with pressure[11]. These demonstrations of pressure-dependent ICR confirm that mechanical forcing can tune the ionic diode behavior of glass nanopores. Building on this discovery, Jubin et al. established a mechanical ionic transistor using conical silica nanopores[85]. Glass nanocapillaries were tested over 0 < ΔP < 1,500 mbar with tip radii ~ 165 ± 15 nm, and the coupled electro-pressure response becomes nonlinear for small ΔP (ΔP < 50 mbar). The conical geometry amplifies local pressure gradients and electric double layer overlap, causing small pressure variations to trigger large conductance changes. These results demonstrate that mechanical sensitivity and output power can be tuned via geometry and surface-charge polarity, turning rigid glass channels into controllable mechanical-electrical converters.
Figure 3. Illustration of bioinspired mechanosensitive ion channels based on glass channels and graphene channels. (A) The single conical glass nanopore and its SEM image. Reproduced with permission[85]. Copyright 2022, National Academy of Sciences. (B) Ion distribution around the pore of the negatively charged glass nanochannel membrane under both positive and negative potentials, with or without ΔP-driven flow. Reproduced with permission[11] Copyright 2011, American Chemical Society; (C) (i) Ion transport through graphene nanopores under the influence of pressure. (ii)TEM image of graphene membrane on 70nm supported SiNx. (iii) Electron diffraction pattern of the graphene membrane. Reproduced with permission[12]. Copyright 2022, The American Association for the Advancement of Science. ΔP: Pressure difference; ΔV: voltage difference; PMMA: poly(methyl methacrylate).
Two-dimensional crystal nanochannels. The narrow channels constructed by two-dimensional crystals such as graphene and hexagonal boron nitride can approach the size of hydrated ions[89-91]. Under such ultimate confinement, ion transport no longer follows simple hydrated-radius sieving, and ions with hydrated diameters larger than the slit height can still permeate[91]. Mouterde et al. investigated ΔP-driven flow through angstrom-scale slits made from graphite and hexagonal boron nitride (h-BN) and discovered molecular streaming with voltage control[89]. In these channels, a small bias voltage strongly amplifies the ΔP-induced streaming current, giving a transistor-like response in which the streaming mobility can be enhanced by up to an order of magnitude when a fraction-of-a-volt potential is applied. The mechanism arises from the coupling between hydrodynamic flow, ion-wall friction, and material-dependent interfacial properties, so that pressure and voltage jointly control ionic transport. This dual-gating mechanism is similar to the multi-parameter control of biological mechanosensitive channels, where membrane tension and membrane potential both modulate channel opening. Furthermore, Jiang et al. observed the effect of pressure on ion conductivity within single-layer graphene nanochannels suspended on a SiNx support [Figure 3C][12]. Under the application of a bias voltage, an increase in ΔP led to a significant enhancement in conductivity, ranging from a few percentage points to over 200%, far exceeding the predictions of the classical linear fluid dynamics theory. Overall, angstrom-scale 2D slits and graphene nanopores show that even atomically 2D crystals can exhibit pronounced mechanosensitive ion transport, via pressure-controlled streaming, voltage-amplified flow, and capacitive ion accumulation. Therefore, these materials can serve as the rigid structural framework of BMSCs.
Carbon nanotubes. Carbon nanotubes (CNTs) provide atomically smooth, cylindrical nanopores with diameters comparable to those of biological ion channels. Small-diameter single-walled CNTs can function as bioinspired ion-channel conduits, where fixed negative charges (e.g., carboxylates) at the tube mouths regulate cation selectivity and support multi-ion conduction at high ionic strength[92]. By embedding multiwalled CNTs with a diameter of approximately 2 nm into silicon nitride membranes, Marcotte et al. investigated ionic transport through the resulting nanochannels under the combined action of pressure and applied voltage[13]. They observed that the ionic conductance depends quadratically on the applied pressure, evidencing mechanically activated ion transport. In summary, electrokinetic modulation provides a powerful and device-friendly route to translate mechanical forcing into ionic/electrical signals. Because the dominant input is typically a pressure difference that drives fluid flow and advects excess charge, the resulting mechanosensitivity is most often expressed as continuous transport regulation rather than a discrete barrier-crossing gating event.
Pore/channel deformation
Pore/Channel deformation nanochannels achieve mechanosensitivity by mechanically reconfiguring the channel geometry - including effective radius, shape, entrance curvature, and local constrictions - under applied loading such as pressure, strain, or bending. In this category, the mechanical input is transduced into ionic readouts mainly through morphological changes of the transport pathway, which then secondarily reshape interfacial ion distributions and resistive/rectifying behavior[14,93].
Silicon nitride nanopores. Silicon nitride (SiN) nanochannels are typically fabricated on suspended films with a thickness of 10-100 nm through electron beam lithography (EBL) and reactive ion etching (RIE) techniques[94,95]. They possess the advantages of compatibility with micro-nano processing, controllable pore size (several tens of nanometers), and excellent mechanical properties[94]. In symmetric SiN nanopores, pressure can simultaneously enlarge the pore and induce ionic current rectification [Figure 4A] by elastically deflecting the membrane and reshaping the pore geometry and entrance region[15]. This proves that even “rigid” inorganic membranes possess enough elastic compliance for nanometer-scale mechanical gating. Beyond bare pores, Xu et al. explored nanoparticle-blocked nanopores in free-standing SiN membranes. In their device, a nanoparticle is trapped near the entrance of a SiN nanopore[96]. The ionic current decreases as external pressure increases, contrary to the usual increase expected from simple streaming. This behavior resembles pressure-induced current reduction in BMSCs.
Figure 4. Illustration of pore deformation nanochannels based on SiN and carbon nanotubes. (A) Mechanosensitive SiN nanochannel membranes. The pore size of the membrane changes under the action of pressure. Reproduced with permission[15]. Copyright 2020, American Chemical Society; (B) The dynamic nano-channel membrane based on CNTs. It can be reversibly switched between the unbent state and the curved state. Reproduced with permission[14]. Copyright 2019, Wiley-VCH. PDMS: Poly(dimethylsiloxane).
Carbon nanotubes mounted in flexible polymers. Beyond changing pore radius, an emerging strategy for pore-deformation mechanosensitive is to program channel asymmetry at the device level by mechanically reshaping the nanochannel curvature. Wang et al. developed a dynamic curvature nanochannel-based membrane [Figure 4B], illustrating how CNTs can be mechano-gated at the device level[14]. In this system, CNT arrays are embedded in an elastomeric support, and bending the membrane changes the local curvature of the nanochannels without significantly altering their radius. The ICR ratio can be inverted when the membrane transitions from an unbent to a curved state, and the switching is reversible under cyclic bending. Carbon-based tubular nanochannels can function as robust, integrable BMSCs, bridging the gap between molecular-scale mechanics and macroscopic iontronic devices.
Block copolymer nanochannels. Block Copolymers (BCPs) provide a powerful bottom-up route to fabricate dense, structurally well-defined nanochannel membranes with tunable mechanics. Microphase separation of amphiphilic BCPs into cylindrical, lamellar, or gyroid morphologies, followed by orientation of conducting domains normal to the membrane plane, yields arrays of parallel channels with diameters typically in the 5-50 nm range and areal densities up to ~ 1011 cm-2[97,98]. Such BCP-derived membranes have been extensively explored as ion-exchange[99], energy conversion[100-102], and sensor[18,103], where the microphase-separated, continuous ion-conducting domains provide efficient ion/molecule transport pathways while the complementary block supplies mechanical robustness. By leveraging the flexibility and elasticity of the BCP framework, the externally applied mechanical stimuli can be transformed into changes in the geometric morphology, wetting state, and energy barrier structure of the nanochannels, thereby simulating the gating behavior of natural mechanically sensitive ion channels in an artificial synthetic system.
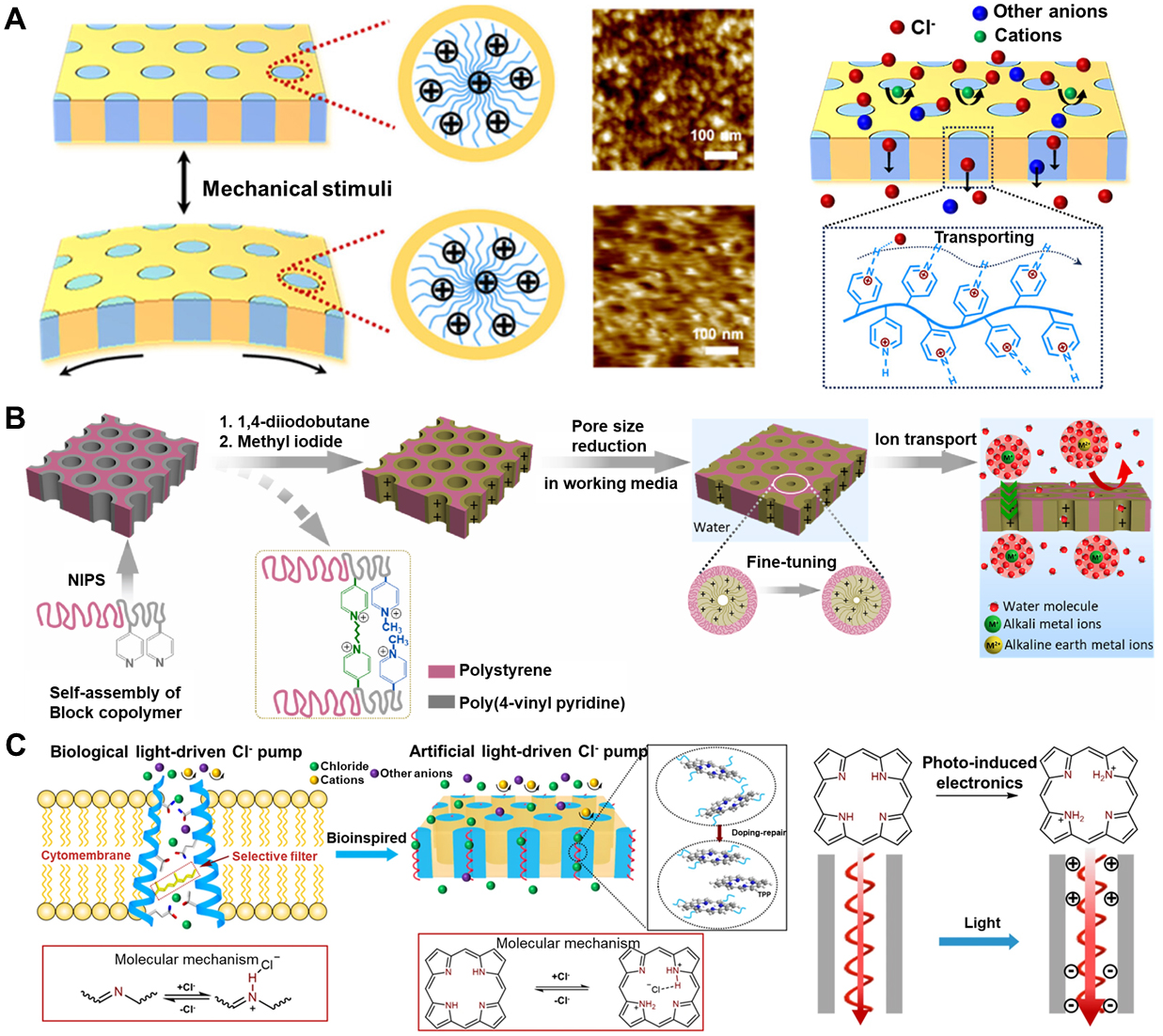
A representative example of truly mechanosensitive polymer nanochannels is the bioinspired elastomeric membrane developed by Li et al.[18] They used the polyisoprene-b-poly4-vinylpyridine (PI-b-P4VP) BCPs via spin coating and self-assembly to form a hexagonally packed cylinder (HPC) structure, where PI forms the continuous phase and P4VP forms the transmembrane hexagonal cylinders [Figure 5A]. The P4VP domains form vertically aligned, chloride-selective nanochannels across the film, while the PI block provides a soft, highly stretchable scaffold. Upon applying small external pressures, the elastomeric PI matrix transduces macroscopic deformation into internal tensile stress, which drives pronounced shape changes of the P4VP channels. The deformation of the nanochannels (rather than a simple change in membrane area) is the reason for the enhanced ion response, which is very similar to the “force-from-lipids” mechanism of natural mechanically sensitive channels.
Figure 5. Schematic diagram of the bioinspired ion channel based on flexible polymer nanochannels. (A) The PI-b-P4VP-based bioinspired mechanosensitive ion channel. Upon mechanical stimulation of the membrane, the P4VP channel undergoes significant deformation. Reproduced with permission[18]. Copyright 2023, American Chemical Society; (B) Illustration of modifying the inner wall of the channel for fine-tuning the pore size. Reproduced with permission[104]. Copyright 2024, Elsevier; (C) The bioinspired photo-responsive polymer ion channels. Reproduced with permission[99]. Copyright 2024, Springer Nature. Light induces the formation of porphyrins inside, generating surface charges that promote the formation of chloride ion channels. PI-b-P4VP: Polyisoprene-b-poly4-vinylpyridine.
Recent studies have exploited the rich chemical versatility of BCPs to achieve responsive control over nanoscale channels [Figure 5B]. Although such control is predominantly triggered by pH[104] or light[99] [Figure 5C] rather than by mechanical force, the BCP framework nonetheless provides a robust and scalable platform for constructing BMSCs, for example, through functionalization of the inner walls with mechanoresponsive molecules.
Molecular conformational gating
Molecular conformational gating primarily relies on the rearrangement of conformational states of supramolecular structures under mechanical stress, thereby directly regulating the coordination of ions or through specific pathways. The basic strategy for constructing self-assembled supramolecular channels involves exploiting a variety of noncovalent interactions, such as hydrogen bonding, electrostatic interactions, metal coordination, host-guest recognition, ion-π interactions, and halogen bonding, to drive the assembly of small molecules into functional transmembrane structures[105]. Such dynamic structures, which can undergo structural rearrangement in response to extremely weak stimuli, offer a particularly attractive route for the construction of BMSCs.
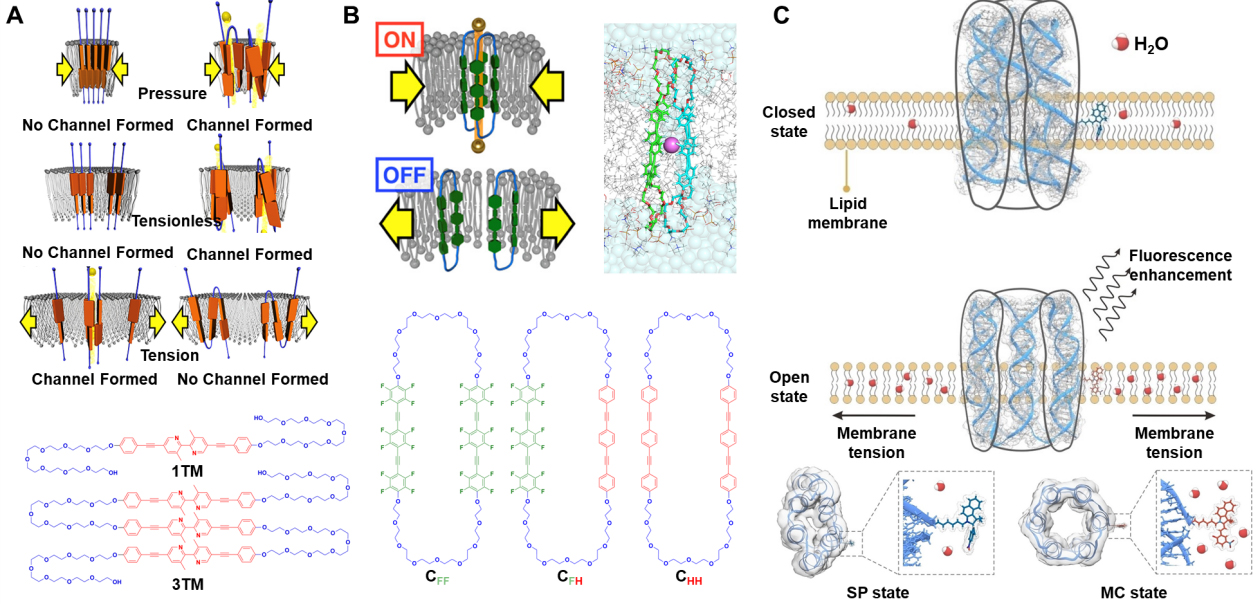
Muraoka et al. constructed a series of amphiphilic molecules bearing one (1TM) or three (3TM) transmembrane molecules by combining oligo(ethylene glycol) chains with aromatic units [Figure 6A][106]. These amphiphiles self-assemble into transmembrane ion channels in lipid bilayers. Subsequently, Sato et al. reported mechanically sensitive K+ channels formed by dimerization or higher-order oligomerization of fluorinated amphiphilic cyclophanes [Figure 6B][16]. By introducing perfluorinated aromatic units, they achieved K+ selectivity through specific C-F···K interactions. Moreover, Zheng et al. developed a bioinspired mechanosensitive channel with a DNA scaffold [Figure 6C][17]. They employed a classical six-helix-bundle DNA nanopore. They introduced three cholesterol groups on the outer surface in the same equatorial plane, forming a “tripod-like” anchoring motif in the lipid bilayer. By further modifying the nanopore with a spiropyran (SP) mechanophore, they transform membrane tension into fluorescence signal. They exploited membrane-tension-induced changes in lipid packing and local microenvironmental polarity around the pore: under increased tension, SP is converted from its weakly emissive, closed-ring SP form into the highly fluorescent, open-ring merocyanine (MC) form.
Figure 6. Illustration of the bioinspired mechanosensitive ion channel based on amphiphilic molecular units and DNA units. (A) The channels of 1TM (left) and 3TM (right) molecules in the pressure, tensionless, and tension states. Reproduced with permission[106]. Copyright 2017, American Chemical Society; (B) The channel structure of CFF and chemical structures of amphiphilic cyclophanes. Reproduced with permission[16]. Copyright 2022, American Chemical Society; (C) The structural deformation of mechanosensitive DNA nanochannels under membrane tension. Reproduced with permission[17]. Copyright 2023, Wiley-VCH. TM: Transmembrane; SP: spiropyran; MC: merocyanine; CFF: fluorinated amphiphilic cyclophane; CFH: partially fluorinated amphiphilic cyclophane; CHH: nonfluorinated.
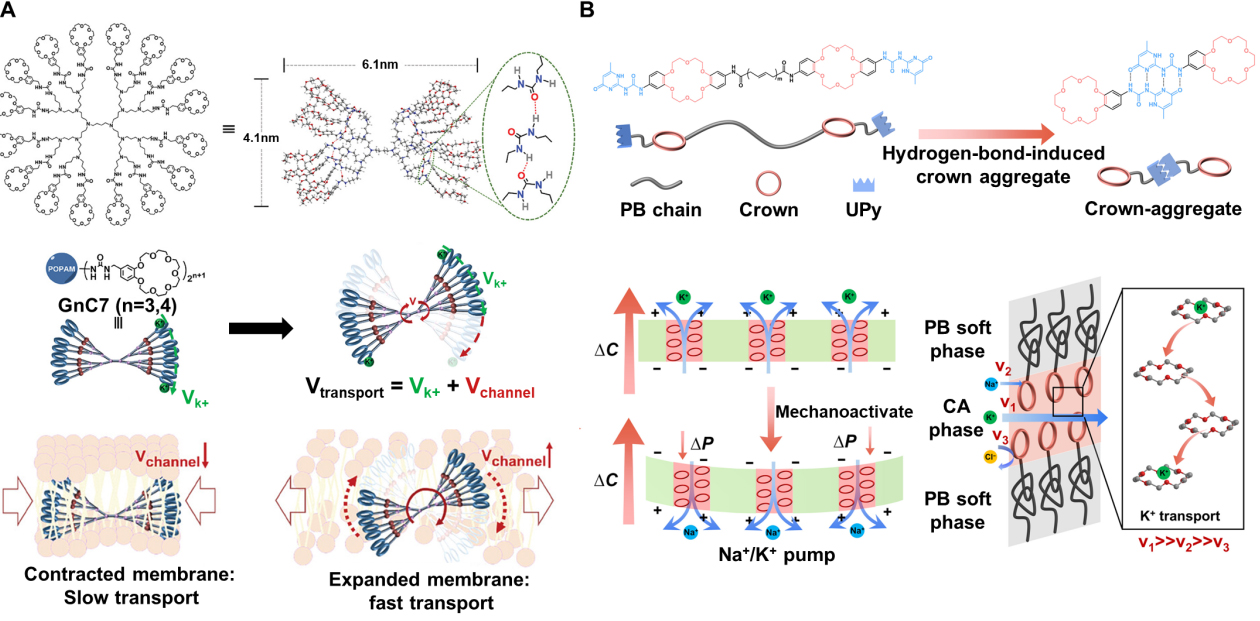
More recently, Wu et al. constructed a mechanically and pH dual-gated butterfly-shaped artificial ion channel (called GnC7) on a poly(propylene imine) (PPI) dendrimer and benzo-21-crown-7 ethers [Figure 7A][19]. In this design, the PPI dendrimer serves as the central scaffold, and each terminal branch is functionalized with a benzo-21-crown-7 ether, collectively forming a multisite K+-coordination array in three-dimensional space. Multiple intramolecular hydrogen bonds and π-π interactions among the crown-bearing arms induce them to bend inward, giving rise to an overall curved, arc-like conformation. Two such arc-shaped units can dimerize within the membrane to form a butterfly-shaped transmembrane structure that spans the lipid bilayer, where the two bent “wings” constitute the coordination regions and the intervening space defines a K+-selective conduction pathway. Owing to this topology, which is both preorganized and mechanically compliant, GnC7 can undergo mechanically driven arc-like rotation within the plane of the membrane. Such rotation alters the relative orientations of the crown-ether coordination sites and modulates the size of the coordination cavity inside the channel, thereby enabling mechanical gating of ion transport. In parallel, Dong et al. constructed a series of bioinspired mechanosensitive channels using pillar[5]arene as a controllably flexible backbone[107]. By varying the length of the alkyl linkers connecting the side chains, they finely adjusted the overall flexibility of the channel framework. In low-curvature, low-tension “resting” membranes, all channels exhibit a certain level of ion transport activity, with the more rigid backbones showing slightly higher baseline conductance. As membrane curvature and tension increase, however, the more flexible channels display a pronounced enhancement in ion transport activity, while the rigid channels are only weakly affected. Li et al. designed telechelic polymers bearing terminal 18-crown-6 and quadruple hydrogen-bond (UPy) units [Figure 7B][108]. The UPy units self-assemble via quadruple hydrogen bonding, creating a dynamic crosslinked network that organizes the crown ethers at the polymer chain ends. In the absence of a ΔP across the membrane, the channel exhibits ultrahigh ion selectivity, with a K+/Na+ selectivity of up to 104.7. When an external ΔP is loaded, the hydrogen-bonded network undergoes reversible reconfiguration, altering the aggregation state of the crown-ether transport pathways. This deformation slows K+ transport while accelerating Na+ transport, thereby changing the direction and rate of the ion flow. As a result, the system successfully recapitulates key features of action-potential propagation in biological neurons.
Figure 7. Illustration of the butterfly-shaped and pillar-shaped bioinspired mechanosensitive ion channel. (A) The mechanosensitive butterfly-shaped K-channel. Mechanical stimulation will accelerate the K+ transport of GnC7. Reproduced with permission[19]. Copyright 2025, Wiley-VCH; (B) The crown ether aggregate membrane for the K channel. During the resting state, K+ is selectively transported. When ΔP is loaded, the direction and rate of the ion flow reverse.[108] Copyright 2026, The American Association for the Advancement of Science. GnC7: Mechanically and pH dual-gated butterfly-shaped artificial ion channel; PB: polybutadiene; CA: crown-aggregate; UPy: terminal 2-ureido-4-pyrimidinone ΔP: pressure difference.
Overall, supramolecular artificial channels can be engineered to convert external stimuli such as membrane tension and curvature into electrochemical or optical signals. This strategy provides an excellent route to construct BMSCs with high selectivity, high sensitivity, and multi-stimuli responsiveness.
To integrate the diverse design routes discussed above - electrokinetic modulation nanochannels, pore-deformation nanochannels, and molecular conformational gating nanochannels - we summarize representative BMSCs in Table 2. At present, the research on BMSCs mainly focuses on using various materials to achieve the functions of natural MSCs, and a unified detection method is lacking. Therefore, we only discuss the materials of BMSCs and the common detection parameters (including bias pressure, bias voltage, current increment, and ion selectivity). The BMSCs constructed using solid inorganic materials usually achieve mechanical responsiveness through electrokinetic modulation or pore deformation. Due to their inherent rigidity, they can form stable channel structures even under large mechanical forces, making them suitable for designing mechanosensitive channels with high activation thresholds. But they usually require a relatively large (100 mbar) depolarizing pressure to be activated, and the resulting current is also much smaller than that of natural MSCs. BMSCs constructed from flexible polymers typically achieve mechanoresponsiveness through pore deformation. BCPs can form relatively stable pore structures through self-assembly. Moreover, due to their own flexibility, these pores can deform under relatively small mechanical stimuli. This endowed them with extremely high sensitivity. Artificial mechanosensitive supramolecular ionic channels often achieve reversible transformation under mechanical force stimulation by utilizing their self-assembly intermolecular forces. These molecules typically assemble within bilayers, where mechanical stimuli are transmitted through the lipids, and they tend to respond primarily to changes in membrane tension rather than to a pressure difference. Because of their small molecular dimensions, these systems typically generate relatively small ionic currents and are therefore more often pursued for treating ion channelopathies, which to some extent limits their utility in developing sensing devices.
Comparison of various bioinspired mechanosensitive ion channels and natural mechanosensitive ion channels
| Channel | Bias pressure | Bias voltage | Current increment | HPTS assay | Ion selectivity | Ref |
| Glass conical nanopores | 213 mbar | 400 mV | ~ 180 pA | N/A | N/A | [11] |
| Graphite crystals | 25 mbar | 50 mV | ~ 5 pA | N/A | N/A | [12] |
| CNT | 400 mbar | 50 mV | ~ 6 pA | N/A | Cation | [13] |
| CNT-PDMS | Curvature (no ΔP specified) | 100 mV | ~ 150 nA | N/A | Cation | [14] |
| SiNx | 1,000 mbar | 1,000 mV | ~ 200 pA | N/A | N/A | [15] |
| Fluorinated foldamer channels | < 16 mN/m | 100 mV | ~ 2 pA | ~ 30% (ΔP) ~ 48% (no ΔP) | K+ | [16] |
| PI-b-P4VP block copolymers | 0.2 mbar | 1,000 mV | 2,460 nA | N/A | Cl- | [18] |
| Butterfly-shaped supramolecular channel | -19 ~ 25 mN/m | N/A | N/A | 44% (ΔP) 28% (no ΔP) | K+ | [19] |
| Pillar[5]arene | Curvature (no ΔP specified) | 100 mV | 2.35 ~ 2.82 pA | 71% (ΔP) ~ 40% (no ΔP) | N/A | [107] |
| 18-crown-6 and quadruple hydrogen-bond unit | 0 ~ 3 mbar | 1,000 mV | ~ 10 μA (0 mbar) ~ 8 μA (3 mbar) | N/A | K+ (no ΔP) Na+ (ΔP) | [108] |
| MscL | 213 mbar | 20 mV | 500 pA | N/A | No | [126] |
| Piezo | 93 mbar | 100 mV | 2 nA | N/A | Cation | [127] |
| TRAAK | 66 mbar | 100 mV | 800 pA | N/A | Cation | [128] |
THE APPLICATION OF BIOINSPIRED MECHANOSENSITIVE ION CHANNELS
The BMSCs can convert the mechanical stimulation acting on the membrane into electrical, chemical, and optical information. This characteristic enables it to demonstrate potential applications in fields [Figure 8] such as disease treatment, highly sensitive mechanical sensing detection, and nanofluidic iontronic memristor.
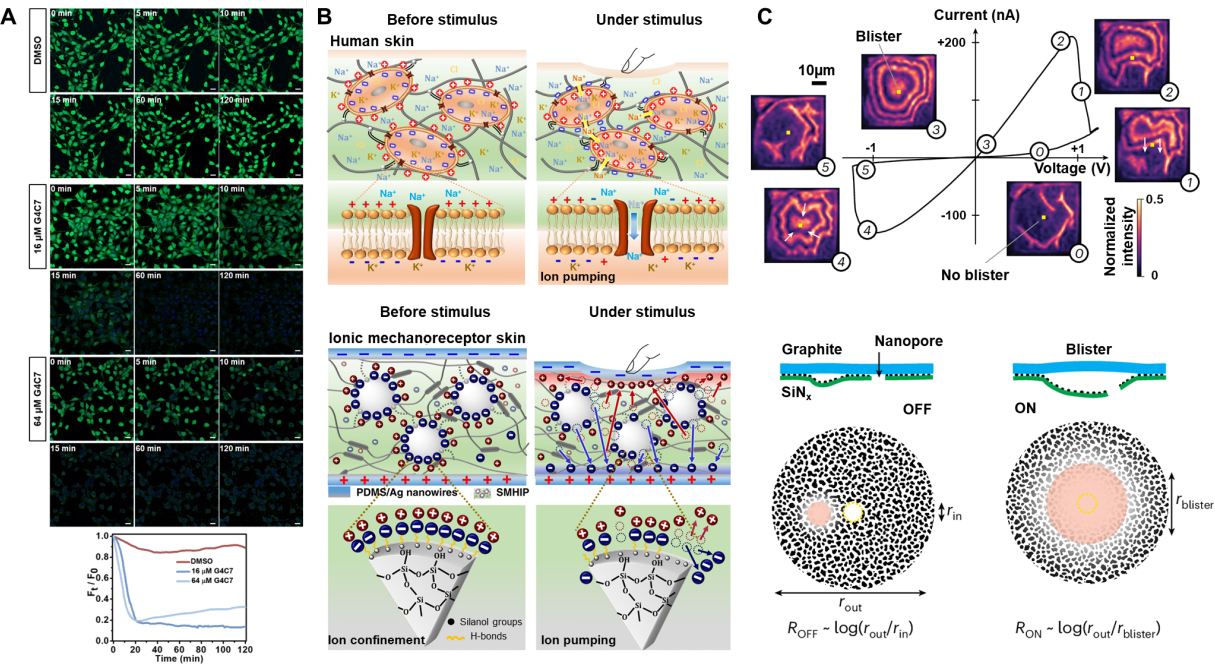
Figure 8. Schematic diagram of the application of bioinspired mechanosensitive ion channels, including promoting apoptosis, artificial skin, and nanofluidic iontronic memristor. (A) Mechanically sensitive butterfly-shaped K+ channels encourage apoptosis in cells by disrupting potassium ion homeostasis. Reproduced with permission[19]. Copyright 2025, Wiley-VCH; (B) Illustration of human skin and Ionic mechanoreceptor skin before and under stimulation. Reproduced with permission[115]. Copyright 2019, Springer Nature; (C) Highly asymmetric channels as the I-V characteristics and schematic diagram of nanofluid memristors. Reproduced with permission[121]. Copyright 2024, Springer Nature. G4C7: Mechanically and pH dual-gated butterfly-shaped artificial ion channel; DMSO: dimethyl sulfoxide; PDMS: poly(dimethylsiloxane); ROFF: conductance ratio in the off state; Ron: conductance ratio in the on state rout: radius of outside; rin: radius of inside; rblister: radius of blister.
Disease treatment
The most direct application of artificial ion channels in biomedicine is to induce cell death by disrupting abnormal ion homeostasis both inside and outside cells. Early studies have demonstrated that small-molecule transporters or artificial channels can serve as “ion drugs”, for instance, by promoting Cl- influx to induce apoptosis of tumor cells[109,110], or by disrupting the cell’s cation balance through Na+/K+ exchange[111], thereby triggering cell death. Mechanically sensitive artificial channels have further developed on this basis, integrating therapeutic ion transport with mechanical signals in the microenvironment. An excellent example is the butterfly-shaped potassium ion channel developed by Wu et al., which features dual gating mechanisms of mechanical and pH[19]. In this system, a dendritic scaffold bearing multiple crown ether units forms a K+-selective transmembrane channel (already discussed in Molecular conformational gating). Membrane deformation and acidified tumor-like conditions cooperatively enhance K+ flux, driving mitochondrial dysfunction and apoptosis in cancer cells. Owing to their ion selectivity and efficient ion transport, ion channels have been developed to treat arrhythmias caused by dysfunctional K+ channels in cardiomyocytes, such as type 2 long QT syndrome (LQT2)[112]. They have also been explored to restore the electrophysiological activity of neurons by inducing action potentials, with potential applications in treating epilepsy and cognitive disorders[113].
Highly sensitive mechanical sensing detection
Mammals encode touch, pressure, and pain using distributed mechanoreceptors coupled to ion channels such as Piezo and various TRP family members[3,114]. The mechanically sensitive ion channel membranes are naturally suitable as mechanical sensing units because they can amplify minute deformations into ion currents. Ionic tactile sensors typically convert mechanical stimuli into electrochemical signals using soft ionic materials, similar to how biological tissues convert external forces into neural impulses. Amoli et al. constructed a synthetic multicellular hybrid ion pump in which hydrogen-bonded ion pairs are dynamically confined on silica microstructures embedded in a thermoplastic polyurethane matrix[115]. Mechanical stimulation can activate this reversible ion pump that is triggered by hydrogen bonds, thereby enabling ultra-sensitive (48.1-5.77 kPa-1) pressure sensing over a wide range (0-135 kPa) at extremely low operating voltages (1 mV). Li et al. constructed high-density Cl--selective elastic nanochannels[18]. These nanochannels maintained stable responses even at a low pressure of 0.2 mbar, amplifying extremely weak mechanical disturbances into measurable current changes[18]. Dobashi et al. showed that pressure gradients across hydrogels produce piezoionic currents whose magnitude and duration are governed by the mobility mismatch between cations and anions[116]. Recently, Zheng et al. were inspired by deep-sea glass sponges and developed an ion-electronic “skin”[117]. This type of skin employs a hydrophobic ionic gel with an open pore structure to ensure that it maintains stable ion channels even under high-pressure conditions. This behavior provides a new strategy for developing highly sensitive mechanical sensing devices.
Nanofluidic iontronic memristor
The memristor can be regarded as a “resistor with memory”. Its instantaneous conductance is not only dependent on the current applied voltage or current, but also on the past excitation history. Confined electrolytes can behave as memristive elements and are influenced by the internal ion distribution, interfacial wettability, and channel morphology[118-121]. Based on this, the concept of “bioinspired nanofluidic iontronics” was proposed[122]. The aim is to achieve similar information processing and storage capabilities as the brain through the ionic flow in nanochannels[122,123]. Emmerich et al. deposited palladium (Pd) onto a SiN membrane containing a single nanopore. By exploiting the formation of island-like Pd nanostructures, they generated an asymmetric ionic channel[121]. Under a positive voltage, liquid blisters were observed to form between the palladium-SiN surface and the graphite surface, and then disappear. The formation and expansion of these liquid blisters led to changes in the ionic conductive pathways, thereby achieving the memory effect. Wei et al. reported a mechano-gated ionic piezoelectric memristor[124]. They constructed a mechanically gated device with adjustable stiffness using a double continuous phase variant gel. And they integrated it with an oscillating ionic memristor, which could generate bipolar excitatory and inhibitory neural-like pulses under the action of pressure. He et al. utilized the reversible deformation of poly(dimethylsiloxane) (PDMS) elastic membranes to regulate the opening and closing of nanopores, enabling the ion conductive state to be directly controlled by pressure[125]. This device utilizes the viscoelastic hysteresis and nanopore confinement effect of the PDMS membrane to achieve the memristive characteristics driven by mechanical force. The ionic conductance of BMSCs is controlled by membrane tension, curvature, or local stress, while the conformational transitions between conformations in BMSCs, based on conformational changes, are slow and have hysteresis. This implies that under periodic electro-mechanical stimulation, BMSCs can exhibit the I-V hysteresis characteristic of a memristor.
CONCLUSION AND OUTLOOK
Natural MSCs, such as MscL, Piezo, TRP, members of the K2P family, and OSCA/TMEM63, achieve highly regulated mechanical gating through mechanisms including force generation from lipids. Inspired by these systems, researchers have begun to extend these principles to artificial channels. Electrokinetic modulation nanochannels, pore deformation nanochannels, and molecular conformational gating nanochannels have been employed to construct BMSCs. These artificial channels can convert extremely small pressure gradients, slight bending, or nanoscale changes in curvature into tunable ionic currents.
However, the field is still in its nascent stage and faces several significant challenges that must be addressed to realize its full potential. (i) Although the existing BMSCs have achieved certain functions of MSCs, they often lack the multi-parameter sensitivity, adaptive kinetics, and regulatory complexity of their biological counterparts. Incorporating stimuli-responsive elements (e.g., mechanophores, photo-switchable groups, or pH-sensitive moieties) into BMSCs could create multi-gated systems that respond to complex biological cues; (ii) Many artificial channels, especially those based on flexible polymers or supramolecular self-assembly, are subject to limitations in their mechanical and chemical stability when operating over a long period of time or when exposed to complex physiological environments. Developing composite membranes that combine the robustness of inorganic frameworks with the flexibility and responsiveness of polymers or supramolecules could enhance stability without sacrificing sensitivity; (iii) The field lacks unified protocols for measuring mechanical sensitivity, gating kinetics, and long-term performance across different platforms, making comparative analysis and optimization difficult. In the future, some reliable detection methods should be developed to measure parameters such as sensitivity, response time, stability, selectivity, operating voltage, signal transduction efficiency, and ionic current; (iv) There are no current reports on the release of BMSCs related to drugs, embedding BMSCs in liposomes and polymers, and enabling them to open channels and release small molecules or ionic drugs under the induced membrane tension changes by ultrasound. This will be an extremely attractive application direction for BMSCs in the future.
Looking ahead, the continued refinement of BMSCs - coupled with advances in fabrication, characterization, and system integration - will unlock transformative applications in healthcare, robotics, computing, and beyond. By harnessing the principles of biological force sensing, these artificial ion channels are poised to become foundational components of the next generation of adaptive, intelligent, and interactive material systems.
DECLARATIONS
Authors’ contributions
Conducted the literature analysis, interpretation, and manuscript writing: Chen, X.; Wang, M.
Conceived the perspective and participated in drafting and critically revising the manuscript: Liu, Y.; Deng, T.; Wang, M.
Availability of data and materials
Not applicable.
Financial support and sponsorship
The authors gratefully acknowledge the financial support from the National Natural Science Foundation of China (No. 52273305) and Fujian Provincial Natural Science Foundation of China (Nos. 2023J05012, 2025J09008).
Conflicts of interest
The authors declare that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
REFERENCES
1. Jin, P.; Jan, L. Y.; Jan, Y. N. Mechanosensitive ion channels: structural features relevant to mechanotransduction mechanisms. Annu. Rev. Neurosci. 2020, 43, 207-29.
2. Beaulieu-Laroche, L.; Christin, M.; Donoghue, A.; et al. TACAN is an ion channel involved in sensing mechanical pain. Cell 2020, 180, 956-967.e17.
3. Kefauver, J. M.; Ward, A. B.; Patapoutian, A. Discoveries in structure and physiology of mechanically activated ion channels. Nature 2020, 587, 567-76.
4. Sukharev, S. I.; Blount, P.; Martinac, B.; Blattner, F. R.; Kung, C. A large-conductance mechanosensitive channel in E. coli encoded by mscL alone. Nature 1994, 368, 265-8.
5. Coste, B.; Mathur, J.; Schmidt, M.; et al. Piezo1 and Piezo2 are essential components of distinct mechanically activated cation channels. Science 2010, 330, 55-60.
6. Nilius, B.; Owsianik, G. The transient receptor potential family of ion channels. Genome. Biol. 2011, 12, 218.
7. Renigunta, V.; Schlichthörl, G.; Daut, J. Much more than a leak: structure and function of K2p-channels. Pflugers. Arch. 2015, 467, 867-94.
8. Murthy, S. E.; Dubin, A. E.; Whitwam, T.; et al. OSCA/TMEM63 are an evolutionarily conserved family of mechanically activated ion channels. Elife 2018, 7.
9. Zhang, M.; Shan, Y.; Cox, C. D.; Pei, D. A mechanical-coupling mechanism in OSCA/TMEM63 channel mechanosensitivity. Nat. Commun. 2023, 14, 3943.
11. Lan, W. J.; Holden, D. A.; White, H. S. Pressure-dependent ion current rectification in conical-shaped glass nanopores. J. Am. Chem. Soc. 2011, 133, 13300-3.
12. Jiang, X.; Zhao, C.; Noh, Y.; et al. Nonlinear electrohydrodynamic ion transport in graphene nanopores. Sci. Adv. 2022, 8, eabj2510.
13. Marcotte, A.; Mouterde, T.; Niguès, A.; Siria, A.; Bocquet, L. Mechanically activated ionic transport across single-digit carbon nanotubes. Nat. Mater. 2020, 19, 1057-61.
14. Wang, M.; Meng, H.; Wang, D.; et al. Dynamic curvature nanochannel-based membrane with anomalous ionic transport behaviors and reversible rectification switch. Adv. Mater. 2019, 31, e1805130.
15. Davis, S. J.; Macha, M.; Chernev, A.; Huang, D. M.; Radenovic, A.; Marion, S. Pressure-induced enlargement and ionic current rectification in symmetric nanopores. Nano. Lett. 2020, 20, 8089-95.
16. Sato, K.; Sasaki, R.; Matsuda, R.; et al. Supramolecular mechanosensitive potassium channel formed by fluorinated amphiphilic cyclophane. J. Am. Chem. Soc. 2022, 144, 11802-9.
17. Zheng, H.; Li, H.; Li, M.; et al. A Membrane tension-responsive mechanosensitive DNA nanomachine. Angew. Chem. Int. Ed. Engl. 2023, 62, e202305896.
18. Li, C.; Liu, P.; Zhi, Y.; et al. Ultra-mechanosensitive chloride ion transport through bioinspired high-density elastomeric nanochannels. J. Am. Chem. Soc. 2023, 145, 19098-106.
19. Wu, Y.; Xu, Q.; Chen, Y.; et al. Mechanosensitive and pH-gated butterfly-shaped artificial ion channel for high-selective K+ transport and cancer cell apoptosis. Adv. Mater. 2025, 37, e2416852.
20. Ranade, S. S.; Syeda, R.; Patapoutian, A. Mechanically activated ion channels. Neuron 2015, 87, 1162-79.
21. Kung, C.; Martinac, B.; Sukharev, S. Mechanosensitive channels in microbes. Annu. Rev. Microbiol. 2010, 64, 313-29.
22. Cox, C. D.; Bavi, N.; Martinac, B. Bacterial mechanosensors. Annu. Rev. Physiol. 2018, 80, 71-93.
23. Kapsalis, C.; Wang, B.; El Mkami, H.; et al. Allosteric activation of an ion channel triggered by modification of mechanosensitive nano-pockets. Nat. Commun. 2019, 10, 4619.
24. Chang, G.; Spencer, R. H.; Lee, A. T.; Barclay, M. T.; Rees, D. C. Structure of the MscL homolog from Mycobacterium tuberculosis: a gated mechanosensitive ion channel. Science 1998, 282, 2220-6.
25. Levina, N.; Tötemeyer, S.; Stokes, N. R.; Louis, P.; Jones, M. A.; Booth, I. R. Protection of Escherichia coli cells against extreme turgor by activation of MscS and MscL mechanosensitive channels: identification of genes required for MscS activity. EMBO. J. 1999, 18, 1730-7.
26. Bass, R. B.; Strop, P.; Barclay, M.; Rees, D. C. Crystal structure of Escherichia coli MscS, a voltage-modulated and mechanosensitive channel. Science 2002, 298, 1582-7.
27. Flegler, V. J.; Rasmussen, T.; Böttcher, B. How functional lipids affect the structure and gating of mechanosensitive MscS-like channels. Int. J. Mol. Sci. 2022, 23, 15071.
28. Pliotas, C.; Dahl, A. C.; Rasmussen, T.; et al. The role of lipids in mechanosensation. Nat. Struct. Mol. Biol. 2015, 22, 991-8.
29. Steinbacher, S.; Bass, R.; Strop, P.; Rees, D. C. Structures of the prokaryotic mechanosensitive channels MscL and MscS. In Current Topics in Membranes, Vol. 58; Academic Press, 2007; pp 1-24.
30. Rasmussen, T.; Flegler, V. J.; Rasmussen, A.; Böttcher, B. Structure of the mechanosensitive channel MscS embedded in the membrane bilayer. J. Mol. Biol. 2019, 431, 3081-90.
31. Reddy, B.; Bavi, N.; Lu, A.; Park, Y.; Perozo, E. Molecular basis of force-from-lipids gating in the mechanosensitive channel MscS. Elife 2019, 8.
32. Fang, X. Z.; Zhou, T.; Xu, J. Q.; et al. Structure, kinetic properties and biological function of mechanosensitive Piezo channels. Cell. Biosci. 2021, 11, 13.
33. Alper, S. L. Genetic diseases of PIEZO1 and PIEZO2 dysfunction. Curr. Top. Membr. 2017, 79, 97-134.
34. Saotome, K.; Murthy, S. E.; Kefauver, J. M.; Whitwam, T.; Patapoutian, A.; Ward, A. B. Structure of the mechanically activated ion channel Piezo1. Nature 2018, 554, 481-6.
35. Ge, J.; Li, W.; Zhao, Q.; et al. Architecture of the mammalian mechanosensitive Piezo1 channel. Nature 2015, 527, 64-9.
36. Zhao, Q.; Zhou, H.; Chi, S.; et al. Structure and mechanogating mechanism of the Piezo1 channel. Nature 2018, 554, 487-92.
37. Jiang, Y.; Yang, X.; Jiang, J.; Xiao, B. Structural designs and mechanogating mechanisms of the mechanosensitive Piezo channels. Trends. Biochem. Sci. 2021, 46, 472-88.
38. Guo, Y. R.; MacKinnon, R. Structure-based membrane dome mechanism for Piezo mechanosensitivity. Elife 2017, 6, e33660.
39. Wang, L.; Zhou, H.; Zhang, M.; et al. Structure and mechanogating of the mammalian tactile channel PIEZO2. Nature 2019, 573, 225-9.
40. Chesler, A. T.; Szczot, M.; Bharucha-Goebel, D.; et al. The role of PIEZO2 in human mechanosensation. N. Engl. J. Med. 2016, 375, 1355-64.
41. Ranade, S. S.; Woo, S. H.; Dubin, A. E.; et al. Piezo2 is the major transducer of mechanical forces for touch sensation in mice. Nature 2014, 516, 121-5.
42. Gnanasambandam, R.; Bae, C.; Gottlieb, P. A.; Sachs, F. Ionic selectivity and permeation properties of human PIEZO1 channels. PLoS. ONE. 2015, 10, e0125503.
43. Liu, S.; Yang, X.; Chen, X.; et al. An intermediate open structure reveals the gating transition of the mechanically activated PIEZO1 channel. Neuron 2025, 113, 590-604.e6.
44. Zhou, Z.; Martinac, B. Mechanisms of PIEZO channel inactivation. Int. J. Mol. Sci. 2023, 24, 14113.
45. Poole, K.; Herget, R.; Lapatsina, L.; Ngo, H. D.; Lewin, G. R. Tuning Piezo ion channels to detect molecular-scale movements relevant for fine touch. Nat. Commun. 2014, 5, 3520.
46. Bae, C.; Sachs, F.; Gottlieb, P. A. The mechanosensitive ion channel Piezo1 is inhibited by the peptide GsMTx4. Biochemistry 2011, 50, 6295-300.
47. Startek, J. B.; Boonen, B.; Talavera, K.; Meseguer, V. TRP channels as sensors of chemically-induced changes in cell membrane mechanical properties. Int. J. Mol. Sci. 2019, 20, 371.
49. Arnadóttir, J.; Chalfie, M. Eukaryotic mechanosensitive channels. Annu. Rev. Biophys. 2010, 39, 111-37.
50. Moparthi, L.; Zygmunt, P. M. Human TRPA1 is an inherently mechanosensitive bilayer-gated ion channel. Cell. Calcium. 2020, 91, 102255.
51. Stanley, S. A.; Kelly, L.; Latcha, K. N.; et al. Bidirectional electromagnetic control of the hypothalamus regulates feeding and metabolism. Nature 2016, 531, 647-50.
52. Servin-Vences, M. R.; Moroni, M.; Lewin, G. R.; Poole, K. Direct measurement of TRPV4 and PIEZO1 activity reveals multiple mechanotransduction pathways in chondrocytes. Elife 2017, 6, e21074.
53. Zheng, W.; Holt, J. R. The Mechanosensory Transduction machinery in inner ear hair Cells. Annu. Rev. Biophys. 2021, 50, 31-51.
54. Wang, Y.; Guo, Y.; Li, G.; et al. The push-to-open mechanism of the tethered mechanosensitive ion channel NompC. Elife 2021, 10, e58388.
55. Brohawn, S. G.; Su, Z.; MacKinnon, R. Mechanosensitivity is mediated directly by the lipid membrane in TRAAK and TREK1 K+ channels. Proc. Natl. Acad. Sci. U. S. A. 2014, 111, 3614-9.
56. Brohawn, S. G. How ion channels sense mechanical force: insights from mechanosensitive K2P channels TRAAK, TREK1, and TREK2. Ann. N. Y. Acad. Sci. 2015, 1352, 20-32.
57. Schmidpeter, P. A. M. Petroff JT, 2. N. D.; Khajoueinejad, L.; et al. Membrane phospholipids control gating of the mechanosensitive potassium leak channel TREK1. Nat. Commun. 2023, 14, 1077.
58. Natale, A. M.; Deal, P. E.; Minor DL, J. R. Structural insights into the mechanisms and pharmacology of K2P potassium channels. J. Mol. Biol. 2021, 433, 166995.
59. Ávalos Prado, P.; Chassot, A. A.; Landra-Willm, A.; Sandoz, G. Regulation of two-pore-domain potassium TREK channels and their involvement in pain perception and migraine. Neurosci. Lett. 2022, 773, 136494.
60. Brohawn, S. G.; del Mármol, J.; MacKinnon, R. Crystal structure of the human K2P TRAAK, a lipid- and mechano-sensitive K+ ion channel. Science 2012, 335, 436-41.
61. Dong, Y. Y.; Pike, A. C.; Mackenzie, A.; et al. K2P channel gating mechanisms revealed by structures of TREK-2 and a complex with Prozac. Science 2015, 347, 1256-9.
62. Aryal, P.; Jarerattanachat, V.; Clausen, M. V.; et al. Bilayer-mediated structural transitions control mechanosensitivity of the TREK-2 K2P channel. Structure 2017, 25, 708-718.e2.
63. Sorum, B.; Docter, T.; Panico, V.; Rietmeijer, R. A.; Brohawn, S. G. Tension activation of mechanosensitive two-pore domain K+ channels TRAAK, TREK-1, and TREK-2. Nat. Commun. 2024, 15, 3142.
64. Miller, A. N.; Long, S. B. Crystal structure of the human two-pore domain potassium channel K2P1. Science 2012, 335, 432-6.
65. Lolicato, M.; Arrigoni, C.; Mori, T.; et al. K2P2.1 (TREK-1)-activator complexes reveal a cryptic selectivity filter binding site. Nature 2017, 547, 364-8.
66. Zheng, W.; Rawson, S.; Shen, Z.; et al. TMEM63 proteins function as monomeric high-threshold mechanosensitive ion channels. Neuron 2023, 111, 3195-3210.e7.
67. Yuan, F.; Yang, H.; Xue, Y.; et al. OSCA1 mediates osmotic-stress-evoked Ca2+ increases vital for osmosensing in Arabidopsis. Nature 2014, 514, 367-71.
68. Chen, G. L.; Li, J. Y.; Chen, X.; et al. Mechanosensitive channels TMEM63A and TMEM63B mediate lung inflation-induced surfactant secretion. J. Clin. Invest. 2024, 134, e174508.
69. Du, H.; Ye, C.; Wu, D.; et al. The cation channel TMEM63B is an osmosensor required for hearing. Cell. Rep. 2020, 31, 107596.
70. Yan, H.; Helman, G.; Murthy, S. E.; et al. Heterozygous variants in the mechanosensitive ion channel TMEM63A result in transient hypomyelination during infancy. Am. J. Hum. Genet. 2019, 105, 996-1004.
71. Han, Y.; Zhou, Z.; Jin, R.; et al. Mechanical activation opens a lipid-lined pore in OSCA ion channels. Nature 2024, 628, 910-8.
72. Maity, K.; Heumann, J. M.; McGrath, A. P.; et al. Cryo-EM structure of OSCA1.2 from Oryza sativa elucidates the mechanical basis of potential membrane hyperosmolality gating. Proc. Natl. Acad. Sci. U. S. A. 2019, 116, 14309-18.
73. Jojoa-Cruz, S.; Saotome, K.; Murthy, S. E.; et al. Cryo-EM structure of the mechanically activated ion channel OSCA1.2. Elife 2018, 7, e41845.
74. Shan, Y.; Zhang, M.; Chen, M.; et al. Activation mechanisms of dimeric mechanosensitive OSCA/TMEM63 channels. Nat. Commun. 2024, 15, 7504.
75. Liu, X.; Wang, J.; Sun, L. Structure of the hyperosmolality-gated calcium-permeable channel OSCA1.2. Nat. Commun. 2018, 9, 5060.
76. Zhang, M.; Wang, D.; Kang, Y.; et al. Structure of the mechanosensitive OSCA channels. Nat. Struct. Mol. Biol. 2018, 25, 850-8.
77. Paulino, C.; Kalienkova, V.; Lam, A. K. M.; Neldner, Y.; Dutzler, R. Activation mechanism of the calcium-activated chloride channel TMEM16A revealed by cryo-EM. Nature 2017, 552, 421-5.
78. Bavi, N.; Cox, C. D.; Nikolaev, Y. A.; Martinac, B. Molecular insights into the force-from-lipids gating of mechanosensitive channels. Curr. Opin. Physiol. 2023, 36, 100706.
79. Lin, S. Y.; Corey, D. P. TRP channels in mechanosensation. Curr. Opin. Neurobiol. 2005, 15, 350-7.
80. Berrier, C.; Pozza, A.; de Lacroix de Lavalette, A.; et al. The purified mechanosensitive channel TREK-1 is directly sensitive to membrane tension. J. Biol. Chem. 2013, 288, 27307-14.
81. Engbers, S.; van Langevelde, P. H.; Hetterscheid, D. G. H.; Klein, J. E. M. N. Discussing the terms biomimetic and bioinspired within bioinorganic chemistry. Inorg. Chem. 2024, 63, 20057-67.
82. Pérez-Mitta, G.; Albesa, A. G.; Trautmann, C.; Toimil-Molares, M. E.; Azzaroni, O. Bioinspired integrated nanosystems based on solid-state nanopores: “iontronic” transduction of biological, chemical and physical stimuli. Chem. Sci. 2017, 8, 890-913.
83. Xin, W.; Jiang, L.; Wen, L. Engineering bio-inspired self-assembled nanochannels for smart ion transport. Angew. Chem. Int. Ed. Engl. 2022, 61, e202207369.
84. van der Heyden, F. H.; Bonthuis, D. J.; Stein, D.; Meyer, C.; Dekker, C. Power generation by pressure-driven transport of ions in nanofluidic channels. Nano. Lett. 2007, 7, 1022-5.
85. Jubin, L.; Poggioli, A.; Siria, A.; Bocquet, L. Dramatic pressure-sensitive ion conduction in conical nanopores. Proc. Natl. Acad. Sci. U. S. A. 2018, 115, 4063-8.
86. Wang, X.; Guo, C.; Su, Z.; et al. Flexible iontronic pressure sensing technology: advanced structural ionic layer. Iontronics 2026, 2, 1.
87. Liu, J.; Kvetny, M.; Feng, J.; et al. Surface charge density determination of single conical nanopores based on normalized ion current rectification. Langmuir 2012, 28, 1588-95.
88. Yang, J.; Su, H.; Lian, C.; Shang, Y.; Liu, H.; Wu, J. Understanding surface charge regulation in silica nanopores. Phys. Chem. Chem. Phys. 2020, 22, 15373-80.
89. Mouterde, T.; Keerthi, A.; Poggioli, A. R.; et al. Molecular streaming and its voltage control in ångström-scale channels. Nature 2019, 567, 87-90.
90. Radha, B.; Esfandiar, A.; Wang, F. C.; et al. Molecular transport through capillaries made with atomic-scale precision. Nature 2016, 538, 222-5.
91. Esfandiar, A.; Radha, B.; Wang, F. C.; et al. Size effect in ion transport through angstrom-scale slits. Science 2017, 358, 511-3.
92. Amiri, H.; Shepard, K. L.; Nuckolls, C.; Hernández Sánchez, R. Single-walled carbon nanotubes: mimics of biological ion channels. Nano. Lett. 2017, 17, 1204-11.
93. Noh, Y.; Aluru, N. R. Ion transport in two-dimensional flexible nanoporous membranes. Nanoscale 2023, 15, 11090-8.
94. Chou, Y. C.; Masih Das, P.; Monos, D. S.; Drndić, M. Lifetime and stability of silicon nitride nanopores and nanopore arrays for ionic measurements. ACS. Nano. 2020, 14, 6715-28.
95. Wang, Y.; Ying, C.; Zhou, W.; de Vreede, L.; Liu, Z.; Tian, J. Fabrication of multiple nanopores in a SiNx membrane via controlled breakdown. Sci. Rep. 2018, 8, 1234.
96. Xu, Y.; Yazbeck, R.; Duan, C. Anomalous mechanosensitive ion transport in nanoparticle-blocked nanopores. J. Chem. Phys. 2021, 154, 224702.
97. Ma, S.; Hou, Y.; Hao, J.; Lin, C.; Zhao, J.; Sui, X. Well-defined nanostructures by block copolymers and mass transport applications in energy conversion. Polymers. (Basel). 2022, 14, 4568.
98. Xiang, L.; Li, Q.; Li, C.; Yang, Q.; Xu, F.; Mai, Y. Block copolymer self-assembly directed synthesis of porous materials with ordered bicontinuous structures and their potential applications. Adv. Mater. 2023, 35, e2207684.
99. Li, C.; Zhai, Y.; Jiang, H.; et al. Bioinspired light-driven chloride pump with helical porphyrin channels. Nat. Commun. 2024, 15, 832.
100. Zhang, Z.; Sui, X.; Li, P.; et al. Ultrathin and ion-selective Janus membranes for high-performance osmotic energy conversion. J. Am. Chem. Soc. 2017, 139, 8905-14.
101. Chen, W.; Xiang, Y.; Kong, X.; Wen, L. Polymer-based membranes for promoting osmotic energy conversion. Giant 2022, 10, 100094.
102. Li, C.; Jiang, H.; Liu, P.; et al. One porphyrin per chain self-assembled helical ion-exchange channels for ultrahigh osmotic energy conversion. J. Am. Chem. Soc. 2022, 144, 9472-8.
103. Wang, J.; Hou, J.; Zhang, H.; Tian, Y.; Jiang, L. Single nanochannel-aptamer-based biosensor for ultrasensitive and selective cocaine detection. ACS. Appl. Mater. Interfaces. 2018, 10, 2033-9.
104. Zhang, Z.; Rahman, M. M.; Ternes, I.; Bajer, B.; Abetz, V. Engineering soft nanochannels in isoporous block copolymer nanofiltration membranes for ion separation. J. Membr. Sci. 2024, 691, 122270.
105. Zheng, S. P.; Huang, L. B.; Sun, Z.; Barboiu, M. Self-assembled artificial ion-channels toward natural selection of functions. Angew. Chem. Int. Ed. Engl. 2021, 60, 566-97.
106. Muraoka, T.; Umetsu, K.; Tabata, K. V.; et al. Mechano-sensitive synthetic ion channels. J. Am. Chem. Soc. 2017, 139, 18016-23.
107. Dong, J.; Zhang, L.; Hu, Y.; Sun, M.; Hou, J. Artificial transmembrane channels displaying mechanosensitivity. Chin. Chem. Lett. 2026, 37, 111420.
108. Li, C.; Chen, C.; Shi, J.; Han, W.; Fu, Q.; Yang, L. Biomimetic action-potential transmission via mechano-gated potassium channels assembled by quadruple hydrogen-bonded crown ethers. Sci. Adv. 2026, 12, eaea6329.
109. Malla, J. A.; Umesh, R. M.; Vijay, A.; Mukherjee, A.; Lahiri, M.; Talukdar, P. Apoptosis-inducing activity of a fluorescent barrel-rosette M+/Cl- channel. Chem. Sci. 2020, 11, 2420-8.
110. Ren, C.; Ding, X.; Roy, A.; et al. A halogen bond-mediated highly active artificial chloride channel with high anticancer activity. Chem. Sci. 2018, 9, 4044-51.
111. Park, S. H.; Hwang, I.; McNaughton, D. A.; et al. Synthetic Na+/K+ exchangers promote apoptosis by disturbing cellular cation homeostasis. Chem 2021, 7, 3325-39.
112. Sun, S.; Xu, Z.; Lin, Z.; et al. A biomimetic ion channel shortens the QT interval of type 2 long QT syndrome through efficient transmembrane transport of potassium ions. Acta. Biomater. 2024, 181, 391-401.
113. Xiao, K.; Sun, Z.; Jin, X.; et al. ERG3 potassium channel-mediated suppression of neuronal intrinsic excitability and prevention of seizure generation in mice. J. Physiol. 2018, 596, 4729-52.
114. Guichard, M.; Thomine, S.; Frachisse, J. M. Mechanotransduction in the spotlight of mechano-sensitive channels. Curr. Opin. Plant. Biol. 2022, 68, 102252.
115. Amoli, V.; Kim, J. S.; Jee, E.; et al. A bioinspired hydrogen bond-triggered ultrasensitive ionic mechanoreceptor skin. Nat. Commun. 2019, 10, 4019.
116. Dobashi, Y.; Yao, D.; Petel, Y.; et al. Piezoionic mechanoreceptors: Force-induced current generation in hydrogels. Science 2022, 376, 502-7.
117. Zheng, Q.; Zhang, D.; Bu, T.; et al. A bioinspired deep-sea iontronic skin for underwater robotic tactile sensing. npj. Flex. Electron. 2025, 10, 8.
118. Xiong, T.; Li, W.; Yu, P.; Mao, L. Fluidic memristor: bringing chemistry to neuromorphic devices. Innovation. (Camb). 2023, 4, 100435.
119. Xie, B.; Xiong, T.; Li, W.; et al. Perspective on nanofluidic memristors: from mechanism to application. Chem. Asian. J. 2022, 17, e202200682.
120. Khan, M. U.; Hassan, B.; Alazzam, A.; Eissa, S.; Mohammad, B. Brain inspired iontronic fluidic memristive and memcapacitive device for self-powered electronics. Microsyst. Nanoeng. 2025, 11, 37.
121. Emmerich, T.; Teng, Y.; Ronceray, N.; et al. Nanofluidic logic with mechano-ionic memristive switches. Nat. Electron. 2024, 7, 271-8.
123. Yu, L.; Li, X.; Luo, C.; et al. Bioinspired nanofluidic iontronics for brain-like computing. Nano. Res. 2023, 17, 503-14.
124. Wei, X.; Wu, Z.; Gao, H.; et al. Mechano-gated iontronic piezomemristor for temporal-tactile neuromorphic plasticity. Nat. Commun. 2025, 16, 1060.
125. He, Y.; Lv, H.; Zhang, Y.; et al. Mechano-gated nanofluidic piezomemristor: elastic nanochannel bridging dynamic pressure modulation and neuromorphic plasticity. ACS. Appl. Mater. Interfaces. 2025, 17, 50292-301.
126. Doerner, J. F.; Febvay, S.; Clapham, D. E. Controlled delivery of bioactive molecules into live cells using the bacterial mechanosensitive channel MscL. Nat. Commun. 2012, 3, 990.
127. Moroni, M.; Servin-Vences, M. R.; Fleischer, R.; Sánchez-Carranza, O.; Lewin, G. R. Voltage gating of mechanosensitive PIEZO channels. Nat. Commun. 2018, 9, 1096.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data











Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.