Atom-mutual-embedding strategy fuses four rings into rare tetrahexacyclic system
Keywords
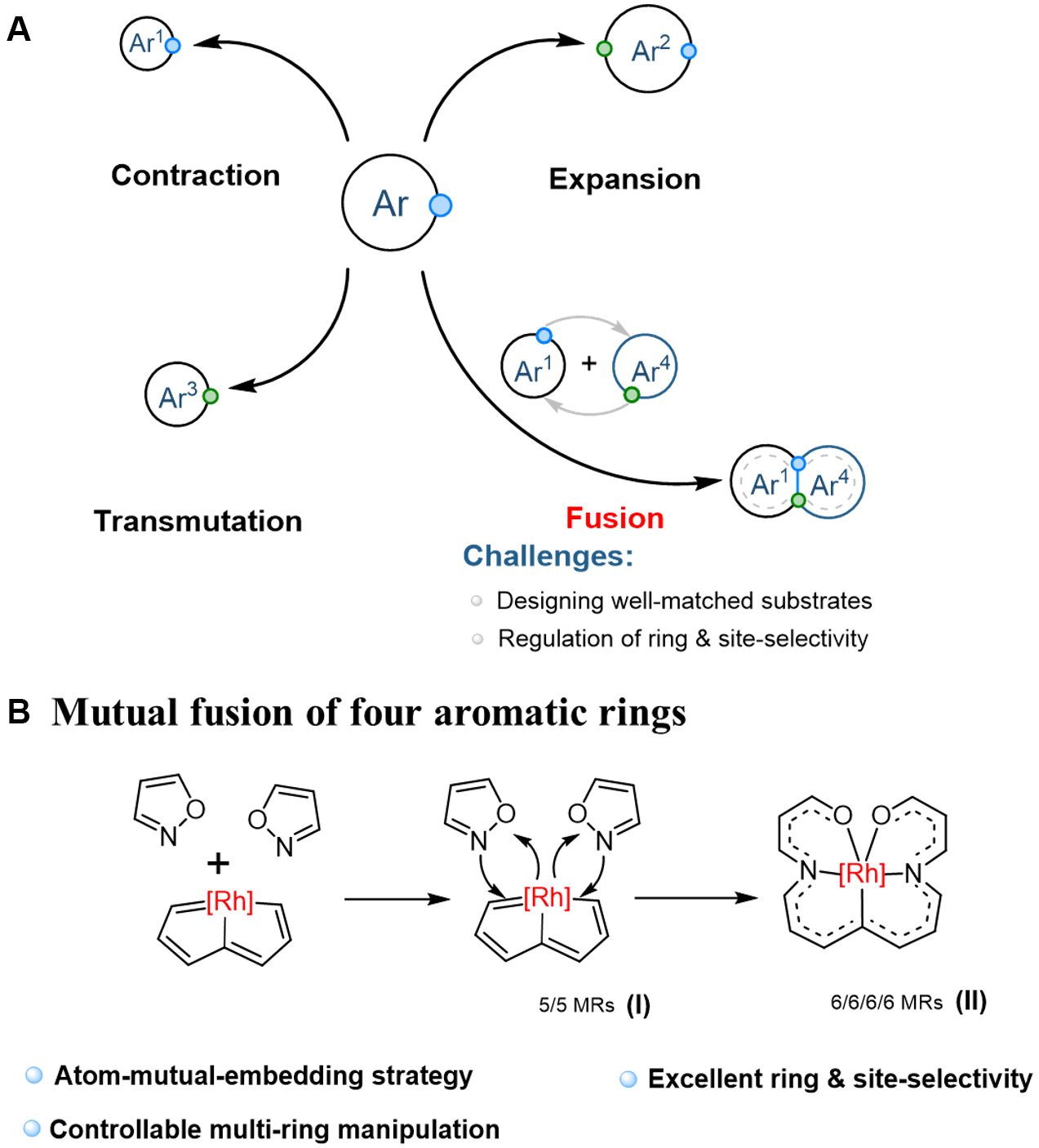
Precise skeletal manipulation of polycyclic aromatic systems remains a formidable synthetic challenge, particularly in achieving controlled ring-opening and reorganization into unified conjugated frameworks. Although significant progress has been made in aromatic ring modification through ring contraction[1,2], expansion[3-15], and single-ring transformations[16-21] [Figure 1A], the precise fusion of multiple aromatic rings with simultaneous control over both connectivity and electronic properties at the atomic level remains a formidable challenge. Xia et al. now address this gap with a mutual-embedding strategy that fuses four 5-membered aromatic rings (two organic isoxazoles and two rhodapentalene-based metalla-aromatics) into a single metal-bridged 6/6/6/6-membered scaffold. Key to this breakthrough is the selective integration of nitrogen atoms into metal-carbon bonds and the formation of a stabilizing metal bridge between isoxazole units. The resulting π-conjugated system exhibits unprecedented thermal stability (up to 160 °C) and enhanced Near Infrared (NIR) properties, surpassing precursor performance [Figure 1B][22].
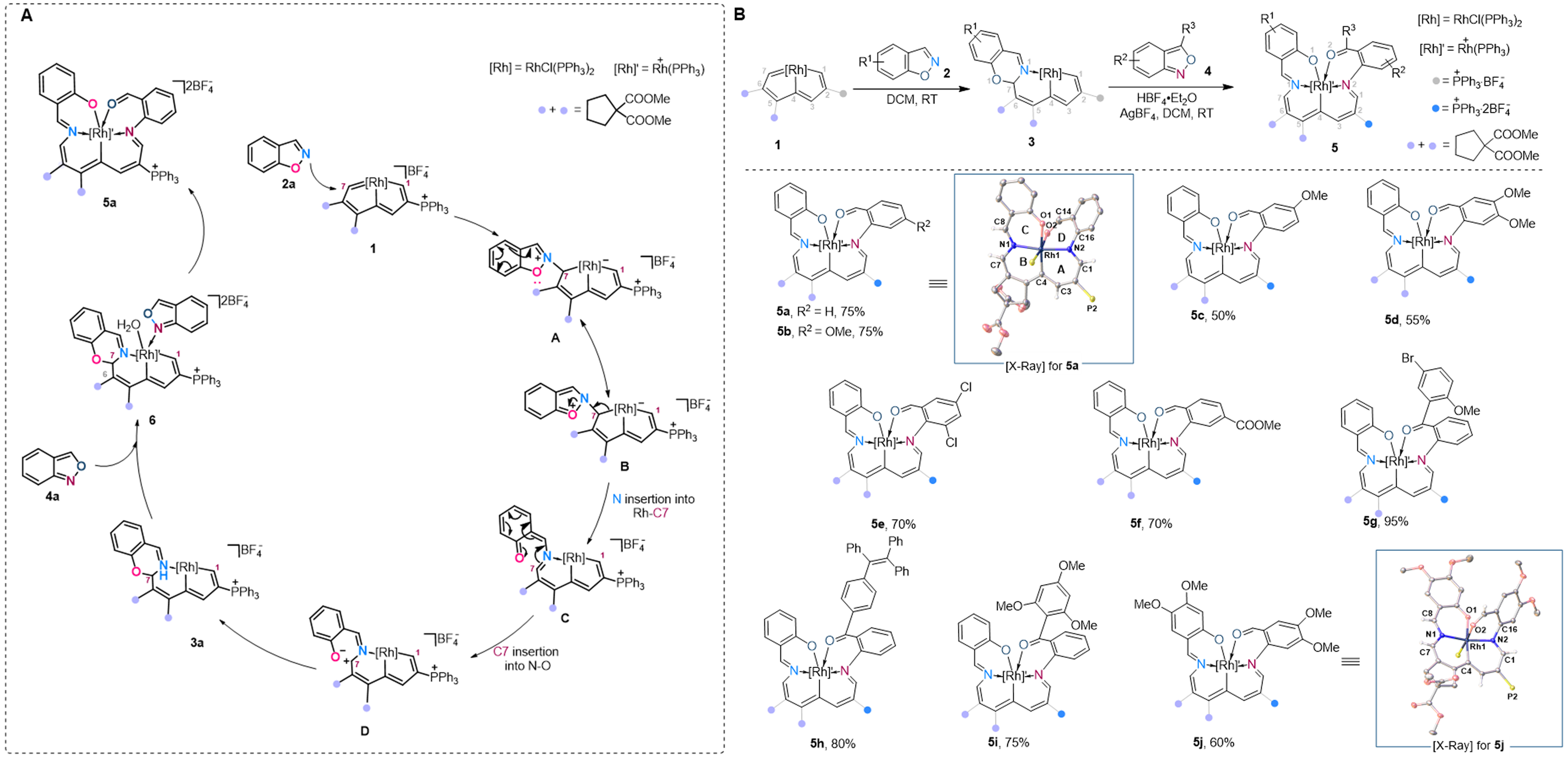
This study elucidates the intricate mechanism of rhodium-mediated polyannulation between benzisoxazoles and rhodapentalenes, which initiates through nucleophilic attack at the C7 position of the metallacycle to form intermediate A. Subsequent electron delocalization, N–O bond cleavage, and rhodium coordination generate imide species C, culminating in aryl migration and cyclization to afford product 3a via a concerted “mutual embedding” process that simultaneously incorporates nitrogen into the Rh-C1 bond and carbon into the N–O linkage. For the construction of more complex 6/6/6/6/6-fused systems, mechanistic investigations reveal that AgBF4-mediated ligand exchange produces pivotal intermediate 6, with subsequent HBF4·Et2O treatment inducing oxygen migration and nitrogen insertion through acid-promoted C–O/N–O bond cleavage of protonated species E. Control experiments demonstrate competing dimerization pathways under specific conditions, highlighting the delicate balance required for selective polycycle formation [Figure 2A]. The transformation exhibits remarkable substrate generality, converting diverse 1,2-benzisoxazole derivatives into corresponding 6/6/6/6-fused products (5a-5j) in 50%-95% yields [Figure 2B].
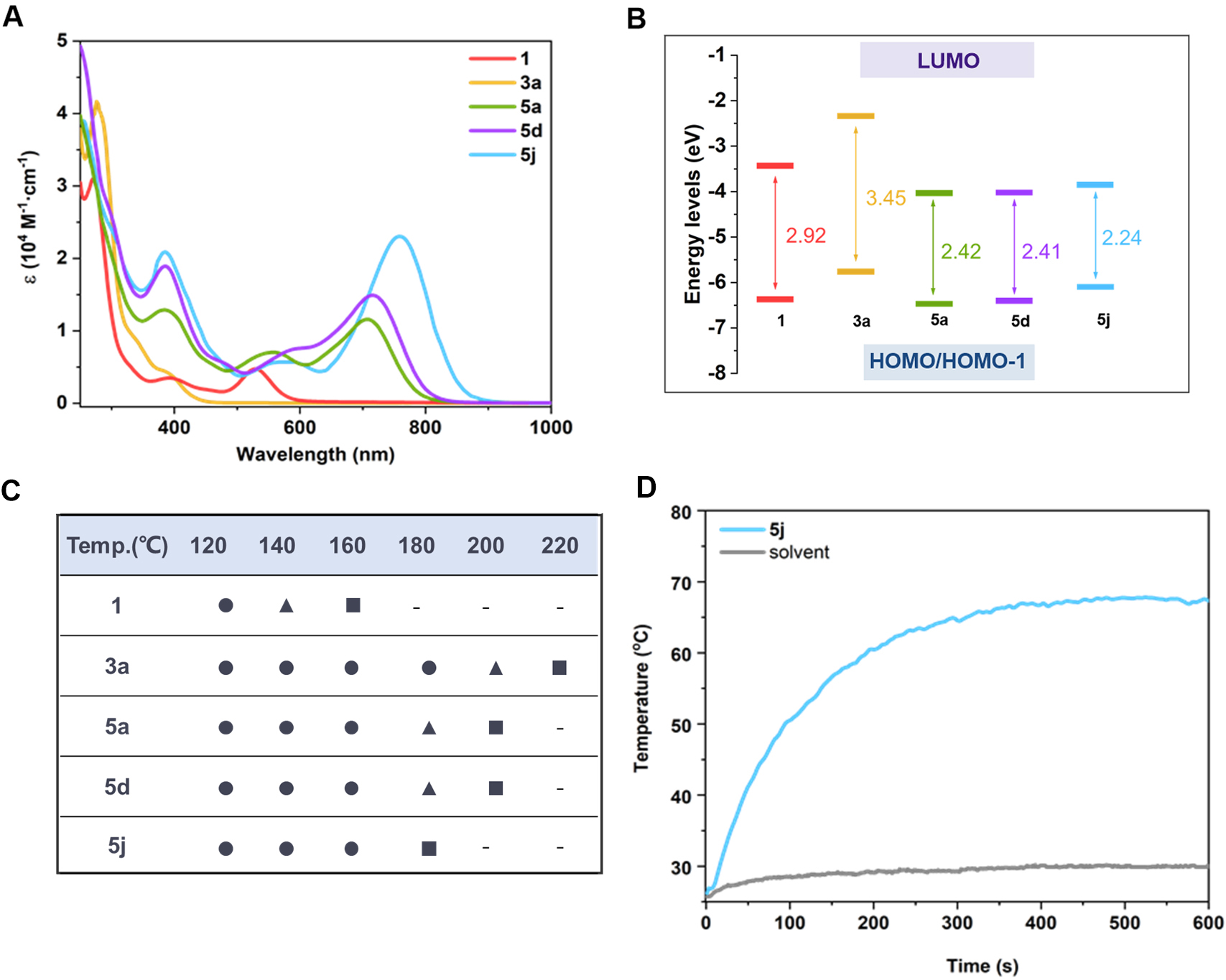
While metallated pyridine frameworks are known, complex 5a represents the first example incorporating two fused metallapyridine units, creating a unique naphthyridine derivative that expands conjugated polycyclic systems. Spectroscopic studies reveal 5a-5j exhibit significant redshifted absorption [Figure 3A] compared to raw materials (1), with Time-Dependent Density Functional Theory calculations confirming narrowed Highest Occupied Molecular Orbital (HOMO)-Lowest Unoccupied Molecular Orbital (LUMO) gaps [Figure 3B]. These compounds exhibit NIR fluorescence, exceptional thermal stability
Figure 3. (A) UV‒Vis-NIR absorption spectra of 1, 3a, 5a, 5d and 5j; (B) Calculated HOMO and LUMO energy levels; (C) Thermal stability of complexes 1, 3a, 5a, 5d and 5j in the solid-state after heating at different temperatures in air for 3 h. ● = stable, ▲ = partly decomposed, ■ = completely decomposed; (D) Photothermal conversion of 5j (0.100 mg/mL in CH3CN) under 808 nm (1.00 W/cm2) laser irradiation. HOMO: Highest Occupied Molecular Orbital; LUMO: Lowest Unoccupied Molecular Orbital. (Figure 3A-D is reproduced with permission[22]. Copyright 2025, American Chemical Society).
In summary, this work by Xia et al. reviews recent advances in the transformation strategies of aromatic compounds, with a particular focus on structural modifications achieved through single-atom skeleton editing and polycyclic fusion strategies. The introduction of aromatic compounds has opened up new possibilities for polycyclic skeleton editing, demonstrating the potential to enhance structural diversity and performance through the fusion of multiple metalloaromatic and organic aromatic systems. Future research should further explore the applications of these strategies in optoelectronic and biomedical fields to enable more extensive functional molecular design.
DECLARATIONS
Authors’ contributions
Writing and revision: Ren, Z.; Wei, L.; Ren, S.
Conceptualization and Project guidance: Pan, Y. M.; Tang, H. T.
Availability of data and materials
Not applicable.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
This work was supported by the National Natural Science Foundation of China (Nos. 22471046 and 22201049).
Conflicts of interest
All authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
REFERENCES
1. Woo, J.; Christian, A. H.; Burgess, S. A.; Jiang, Y.; Mansoor, U. F.; Levin, M. D. Scaffold hopping by net photochemical carbon deletion of azaarenes. Science 2022, 376, 527-32.
2. Bartholomew, G. L.; Carpaneto, F.; Sarpong, R. Skeletal editing of pyrimidines to pyrazoles by formal carbon deletion. J. Am. Chem. Soc. 2022, 144, 22309-15.
3. He, L.; Qu, B.; Xiao, M.; et al. Synthesis of chiral spiro -indenes via Pd-catalyzed asymmetric (4 + 2) dipolar cyclization. Chem. Synth. 2024, 4, 53.
4. Reisenbauer, J. C.; Green, O.; Franchino, A.; Finkelstein, P.; Morandi, B. Late-stage diversification of indole skeletons through nitrogen atom insertion. Science 2022, 377, 1104-9.
5. Mykura, R.; Sánchez-Bento, R.; Matador, E.; et al. Synthesis of polysubstituted azepanes by dearomative ring expansion of nitroarenes. Nat. Chem. 2024, 16, 771-9.
6. Liu, S.; Yang, Y.; Song, Q.; et al. Tunable molecular editing of indoles with fluoroalkyl carbenes. Nat. Chem. 2024, 16, 988-97.
7. Wu, F. P.; Chintawar, C. C.; Lalisse, R.; et al. Ring expansion of indene by photoredox-enabled functionalized carbon-atom insertion. Nat. Catal. 2024, 7, 242-51.
8. Hyland, E. E.; Kelly, P. Q.; McKillop, A. M.; Dherange, B. D.; Levin, M. D. Unified access to pyrimidines and quinazolines enabled by n-n cleaving carbon atom insertion. J. Am. Chem. Soc. 2022, 144, 19258-64.
9. Ouyang, H.; Peng, M.; Song, K.; Wang, S.; Gao, H.; Tan, B. Synthesis of metal-phenanthroline-modified hypercrosslinked polymer for enhanced CO2 capture and conversion via chemical and photocatalytic methods under ambient conditions. Chem. Synth. 2024, 4, 50.
10. Ma, D.; Martin, B. S.; Gallagher, K. S.; Saito, T.; Dai, M. One-carbon insertion and polarity inversion enabled a pyrrole strategy to the total syntheses of pyridine-containing Lycopodium alkaloids: complanadine A and lycodine. J. Am. Chem. Soc. 2021, 143, 16383-7.
11. Wang, J.; Lu, H.; He, Y.; Jing, C.; Wei, H. Cobalt-catalyzed nitrogen atom insertion in arylcycloalkenes. J. Am. Chem. Soc. 2022, 144, 22433-9.
12. He, G.; Chen, X.; Xia, S.; Zhong, G.; Yang, L. Enantioselective synthesis of 6-(Indole-2-yl)-3,4-dihydropyran-2-one skeletons by N-Heterocyclic carbene-catalyzed asymmetric [3 + 3] cycloaddition of α-bromocinnamaldehyde. Chem. Synth. 2023, 3, 35.
13. Ren, S.; Lin, S.; Ren, Z.; et al. Single-site palladium “microreactor” for carbon dioxide coupling/cyclization reactions. ACS. Sustainable. Chem. Eng. 2025, 13, 2001-10.
14. Zhang, X.; Song, Q.; Liu, S.; et al. Asymmetric dearomative single-atom skeletal editing of indoles and pyrroles. Nat. Chem. 2025, 17, 215-25.
15. Wang, T.; Shen, B.; Chen, X.; Wan, Q.; Yu, P.; Li, P. Organocatalytic regio- and enantioselective formal [4 + 2]-annulation of chiral nitrogen-containing dipoles. Chem. Synth. 2023, 3, 9.
16. Woo, J.; Stein, C.; Christian, A. H.; Levin, M. D. Carbon-to-nitrogen single-atom transmutation of azaarenes. Nature 2023, 623, 77-82.
17. Pearson, T. J.; Shimazumi, R.; Driscoll, J. L.; Dherange, B. D.; Park, D. I.; Levin, M. D. Aromatic nitrogen scanning by ipso-selective nitrene internalization. Science 2023, 381, 1474-9.
18. Patel, S. C.; Burns, N. Z. Conversion of aryl azides to aminopyridines. J. Am. Chem. Soc. 2022, 144, 17797-802.
19. Cheng, Q.; Bhattacharya, D.; Haring, M.; Cao, H.; Mück-Lichtenfeld, C.; Studer, A. Skeletal editing of pyridines through atom-pair swap from CN to CC. Nat. Chem. 2024, 16, 741-8.
20. Uhlenbruck, B. J. H.; Josephitis, C. M.; de, Lescure. L.; Paton, R. S.; McNally, A. A deconstruction-reconstruction strategy for pyrimidine diversification. Nature 2024, 631, 87-93.
21. Kim, D.; You, J.; Lee, D. H.; Hong, H.; Kim, D.; Park, Y. Photocatalytic furan-to-pyrrole conversion. Science 2024, 386, 99-105.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data

















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.