StableOx-Cat agent: an AI agent for exploring stable metal oxide electrocatalysts
Abstract
We introduce StableOx-Cat, an artificial intelligence (AI)-agent framework that enables systematic and reliable exploration of stable metal oxide (MO) electrocatalysts via a unified natural-language interface. StableOx-Cat integrates a large language model (LLM) for intent understanding and task orchestration with deterministic, physics-based analysis tools for electrocatalysis evaluation. User queries expressed in natural language are automatically parsed into structured actions, including database statistics, bulk thermodynamic stability screening based on energy-above-hull criteria, and aqueous electrochemical stability analysis under user-defined pH values and electrochemical potential windows. By applying the physical criteria to screen the stable MO electrocatalysts, StableOx-Cat avoids hallucinations and ensures a physically based stability analysis. This Agent enables the assessment of aqueous electrochemical stability across a wide range of reactions, with applied potentials spanning -2 to 2 V versus standard hydrogen electrode and pH values ranging from 0 to 14. Representative use cases demonstrate how StableOx-Cat enables flexible stability screening of MOs under both thermodynamic and aqueous environments. In addition, the agent architecture supports integration with different LLMs for task execution and query parsing. Overall, StableOx-Cat provides an accessible platform for stability-oriented materials exploration, offering a practical pathway to accelerate the discovery of experimentally relevant MO electrocatalysts for electrochemical applications, and can be generalized to other classes of electrocatalysts, such as alloys, metal nitrides, and carbides.
Keywords
INTRODUCTION
Metal oxides (MOs) have attracted growing interest as electrocatalysts due to their abundance on Earth, structural diversity, and tunable chemical properties[1,2]. This broad materials class includes both noble and non-noble MOs. Among them, noble MOs exhibit high electrocatalytic performance, such as RuO2[3,4], which is a well-established catalyst for the oxygen evolution reaction (OER). However, their high cost and limited availability constrain large-scale applications. As a result, considerable effort has been directed toward identifying low-cost MO electrocatalysts[5]. Nevertheless, given the vast compositional and structural space of MOs, conventional trial-and-error approaches remain time-consuming and inefficient, underscoring the need for more efficient strategies to accelerate the discovery of stable, high-performance MO electrocatalysts.
High-throughput first-principles calculations have enabled systematic physical assessments across a wide range of materials. Large-scale computational materials databases, such as the Materials Project (next-gen.materialsproject.org)[6] and AFLOW (Automatic Flow, aflowlib.org)[7], provide computed bulk crystal structures, thermodynamic and physicochemical properties for diverse inorganic compounds, while crystallographic repositories, including the Inorganic Crystal Structure Database (ICSD: icsd.fiz-karlsruhe.de)[8], compile detailed structure information of all experimentally reported crystal structure data. The Digital Catalysis Platform (DigCat: www.digcat.org)[9] further integrates more than 400,000 experimental electrocatalyst data entries and more than 400,000 computational surface data entries. These open databases provide extensive collections of materials data and typically expose application programming interfaces (APIs)[10] or dedicated data-query interfaces[9] to support efficient data access and analysis. The Pymatgen Python library[11] can be used to access the Materials Project API[10] and was designed to retrieve information from thermodynamic phase diagrams[12-14] and bulk Pourbaix diagrams[15-17] in the Materials Project, enabling a systematic assessment of both thermodynamic stability and aqueous electrochemical stability of MOs. Using data infrastructures from the Materials Project and DigCat, Sb2WO6[18] has been explored as a catalyst for oxygen reduction reactions (ORRs) under acidic and alkaline conditions, and RbSbWO6[19] as a bifunctional electrocatalyst for OER and HER in acidic media.
However, effective use of these data infrastructures still poses a significant barrier for many materials scientists, as it typically requires interdisciplinary expertise in programming, data processing, and domain-specific materials knowledge to develop custom scripts and manage data pipelines. With the rapid advancement of large language models (LLMs), conversational question-answering has emerged as a powerful paradigm for human-computer interaction. Nevertheless, when applied directly to materials research, LLMs may produce unreliable or hallucinatory outputs due to the absence of embedded, domain-specific physical knowledge and executable workflows. To mitigate this limitation, integrating materials knowledge with automated, domain-informed code execution within an artificial intelligence (AI) Agent framework offers a promising pathway to lower the entry barrier for materials exploration[20]. Several domain-specific agents have been developed, including AI Agents for hydrogen storage materials[21], metal-organic frameworks (MOFs)[22], and solid-state batteries[23], demonstrating the potential of Agent-based systems to bridge natural language interaction with reliable materials data analysis. Building on these advances, the development of a dedicated agent for assessing MO stability enables users to efficiently query and identify potentially stable MO electrocatalysts under diverse electrochemical reaction conditions through a unified question-answering interface.
In this context, we present the StableOx-Cat Agent: an AI Agent for stable MO electrocatalysts, designed to provide a unified, natural-language-driven interface for searching and analyzing stable MO materials relevant to electrocatalytic applications. By systematically collecting data from public materials databases, compiling analysis codes, and coupling them with LLM-driven reasoning and tool-calling capabilities, StableOx-Cat Agent allows users to interact through natural language queries. Users can perform different actions, including: (i) checking the MOs in the database; (ii) identifying thermodynamically stable MOs; (iii) determining electrochemically stable MOs under specified aqueous conditions; (iv) performing element-based filtering of stable MOs; and (v) evaluating whether user-specified MOs are stable. This conversational, Agent-based design transforms complex, script-based workflows into an accessible, flexible interface, enabling efficient, user-friendly exploration of stable MO electrocatalysts for electrochemical applications.
METHODS
LLM selection and prompt engineering
For the LLMs, we accessed the DeepSeek-R1-Distill-Qwen-7B via the SiliconFlow API and also tested the gemini-3-flash-preview on Google.com. Prompt engineering plays a critical role in enabling LLMs to support task-specific interactions reliably. It is essential to design effective prompts to ensure that user intent is accurately translated into actionable instructions. We implemented a dedicated set of prompts, integrated with the LLM via the LangChain framework, forming the interaction backbone of the StableOx-Cat Agent. The routing system operates within a predefined action space consisting of five discrete operations: dataset size retrieval (count_metal_oxides), thermodynamic stability filtering (thermodynamic_stable), electrochemical stability filtering (list_stable), composition stability evaluation (is_thermo_stable), and element-based dataset filtering (filter_by_elements). Each action is associated with a strictly defined key schema specifying required and optional parameters.
Thermodynamic filtering is executed by defining the energy above the hull (Ehull) values. When specified, the threshold is extracted as a floating-point parameter; otherwise, a default of 0.0 eV is applied. Element constraints are normalized under a strict schema: individual elements must conform to valid chemical symbols, while element groups are restricted to predefined categories, such as “alkali”: {“Li”, “Na”, “K”, “Rb”, “Cs”, “Fr”}, “alkaline_earth”: {“Be”, “Mg”, “Ca”, “Sr”, “Ba”, “Ra”}, “noble”: {“Au”, “Ag”, “Pt”, “Pd”, “Ir”, “Rh”, “Ru”, “Os”}, Automatic inference of representative elements is prohibited to prevent unintended bias.
A hierarchical intent-resolution strategy was implemented to guarantee deterministic behavior. Electrochemical contexts [e.g., ORR, hydrogen evolution reaction (HER), OER, Pourbaix diagrams, acidic/alkaline conditions, pH] trigger a hard override rule that directs the action to “list_stable”, preventing misclassification as a thermodynamic query. Formula-based precedence is defined such that explicit compositions are treated as stability checks unless electrochemical conditions are specified. In the absence of electrochemical reaction keywords, thermodynamic-related actions are used to filter. Electrochemical parameter extraction follows strict normalization rules. pH values are mapped directly from textual descriptors (acidic: pH = 0, neutral: pH = 7, alkaline/basic: pH = 14), and reaction-specific voltage windows are fixed according to canonical ranges[24] (ORR: 0.6-1.0 V vs. the standard hydrogen electrode (SHE); OER: 1.23-2.0 V vs. SHE; HER: -0.8-0.0 V vs. SHE; propylene electrooxidation: 2.0 V vs. SHE). Explicit user-provided voltages override default windows and are automatically sorted. This ensures reproducible electrochemical boundary conditions.
Materials data sources
The Agent’s actions are executed using a local database containing comprehensive information on MOs, including Materials Project and ICSD identifiers, structural information, thermodynamic properties, and electrochemical data on aqueous stability. These data were pre-extracted from the Materials Project[6] database in its latest version (v2025.09.25) and deployed in a local database to support efficient querying and reproducible analysis. To obtain a wide range of possible MO compositions, we defined the conditions to include elements O and metallic elements, while discarding non-metallic elements, metalloids, and radioactive elements, resulting in 35,817 MOs for statistical analysis of both total and thermodynamically stable MOs. The dataset includes the Ehull for each compound, allowing users to define thermodynamic stability using a specified Ehull threshold (default = 0 eV). Under the default criterion, 7,461 thermodynamically stable MOs were identified, including 1,502 materials with Ehull = 0 eV and 5,959 materials associated with ICSD entries, corresponding to an ICSD coverage of ~79.9%. It should be noted that materials with reported ICSD entries are considered thermodynamically stable in this study, regardless of their calculated Ehull values, as experimental evidence indicates their synthesizability. For aqueous stability analysis, these 7,461 thermodynamically stable materials (Ehull = 0 or has ICSD ID) were used to extract the Gibbs free energy difference (ΔGpbx) and phase stability information across electrochemical potentials ranging from -2 to 2 V (vs. SHE) with an interval of 0.01 V, sampled at 15 discrete pH values. This design avoids repeated online database queries, improves computational efficiency, and ensures full reproducibility of all results.
Electrochemical stability analysis via MCP and user interface
Previous studies[16,18,19] have shown that materials with ΔGpbx values smaller than 0.5 eV·atom-1 can be experimentally observed as stable. Therefore, a threshold of ΔGpbx < 0.5 eV·atom-1[16] is commonly adopted as a criterion for identifying materials that may remain stable in aqueous environments. In addition, the presence of a solid-phase domain in the bulk Pourbaix diagram is required to ensure that the material remains thermodynamically stable under the specified electrochemical conditions. Electrochemical stability analysis (i.e., list_stable action) was encapsulated as a standalone Model Context Protocol (MCP) tool. Given a pH value and a potential window (vs. SHE), the MCP module is called to evaluate ΔGpbx and the existing phases across the specified potential range, and to identify stable MOs or determine whether the user-defined MOs are stable. The user interface was implemented using Chainlit (https://docs.chainlit.io), providing an interactive conversational environment. Users can submit natural-language queries and browse results directly in the interface.
RESULTS AND DISCUSSION
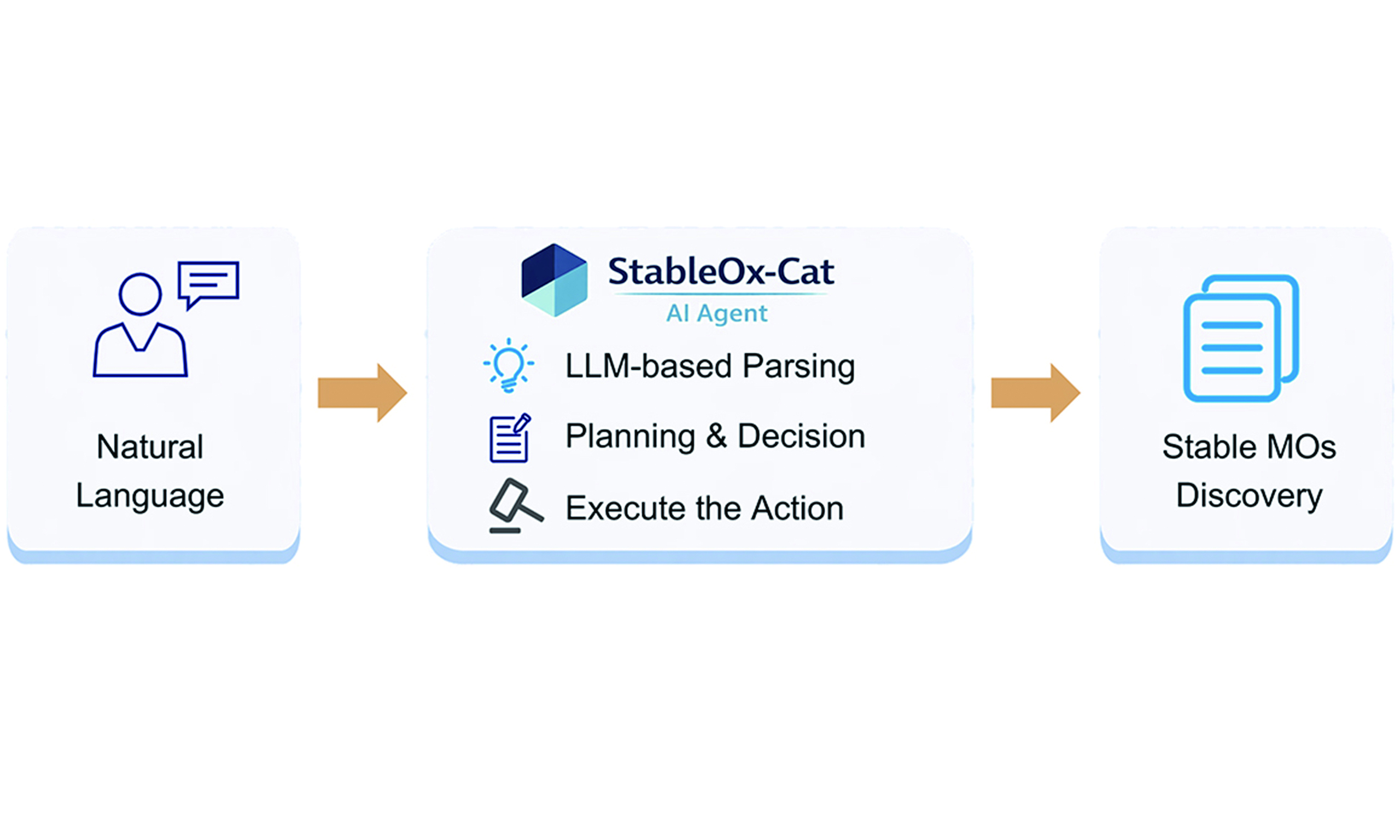
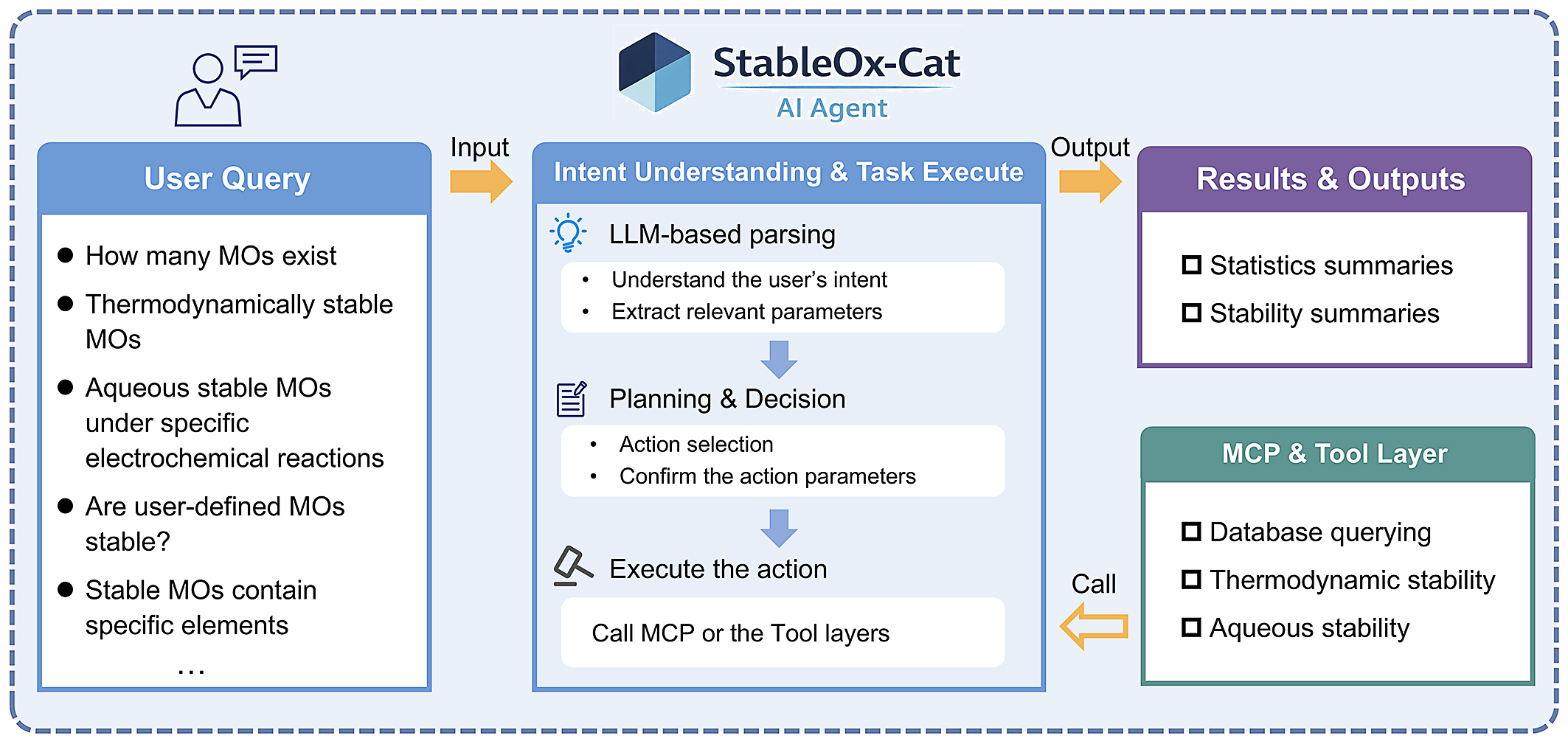
An interactive agent, termed StableOx-Cat, was developed to enable flexible querying and analysis of MO stability under thermodynamic and electrochemical conditions. The system integrates an LLM, guided by carefully designed prompts (details provided in the Methods section), for natural-language intent understanding, with deterministic data-processing modules for database querying and physics-based stability evaluation. Figure 1 illustrates the overall architecture of the StableOx-Cat agent. User queries expressed in natural language, such as the number of known MOs, thermodynamic stability, or aqueous stability under specific electrochemical conditions, are first parsed by an LLM to interpret the user’s intent and extract relevant parameters. Then, the agent plans tasks, makes decisions, dynamically selects appropriate action modules, and confirms the parameters. Finally, the MCP and tool layers execute the action, and the output is obtained (see the video in the Supplementary Materials). StableOx-Cat provides electrocatalysis scientists with an easy-to-use question-answering tool for exploring stable MOs in complex electrochemical environments.
Figure 1. Overall architecture of the StableOx-Cat AI Agent. AI: Artificial intelligence; MOs: metal oxides; MCP: Model Context Protocol.
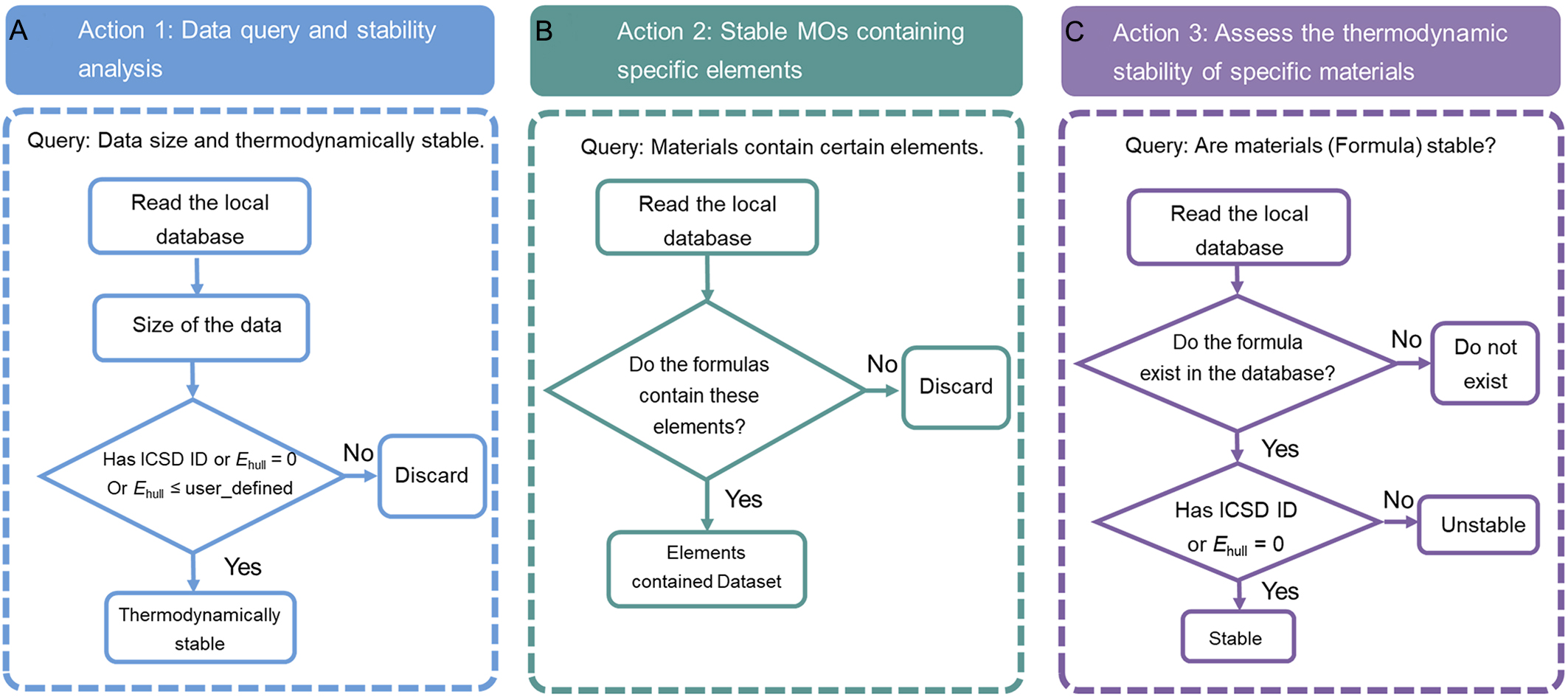
Figure 2 illustrates the functional architecture for analyzing thermodynamically stable MOs, organized into three primary action modules. Action 1 [Figure 2A] is responsible for data retrieval and statistical analysis related to thermodynamic stability. This module directly queries the locally stored database (See the METHODS section) and calculates the dataset size, i.e., the total number of MOs. For thermodynamic stability assessment, MOs are classified as stable based on two criteria: (i) MOs with an ICSD ID are considered stable, as they have been experimentally synthesized and reported; and (ii) MOs with an Ehull lower than a user-defined threshold are also regarded as thermodynamically stable. If the user does not specify a threshold, a default value of Ehull = 0 eV is used. Action 2 [Figure 2B] provides element-based statistical analysis, enabling users to summarize MOs containing specific elements and analyze the associated dataset. This module reads the local database and converts each MO’s composition into a list of constituent elements. The extracted element information is then matched against user-defined elements, allowing filtering by a single metal element or multiple elements for further statistical analysis. Action 3 [Figure 2C] is dedicated to the thermodynamic stability assessment of user-specified materials. This module first queries the local dataset to determine whether the target MO exists in the database. If the MO is not found, the system returns a notification indicating that the MO does not exist in the dataset and prompts the user to verify the input. If the MO is present, the module evaluates its thermodynamic stability based on predefined criteria. Specifically, MOs with an ICSD ID are considered experimentally validated and therefore stable. In addition, a strict thermodynamic condition is applied, with Ehull = 0 eV. When the stability criteria are met, the system returns the relevant material information to the user.
Figure 2. Functional architecture for the analysis of thermodynamically stable MOs, including (A) data query and stability analysis, (B) stable MOs containing specific elements, and (C) assessing the thermodynamic stability for specific MOs. MOs: Metal oxides; ICSD: Inorganic Crystal Structure Database; ID: identifier.
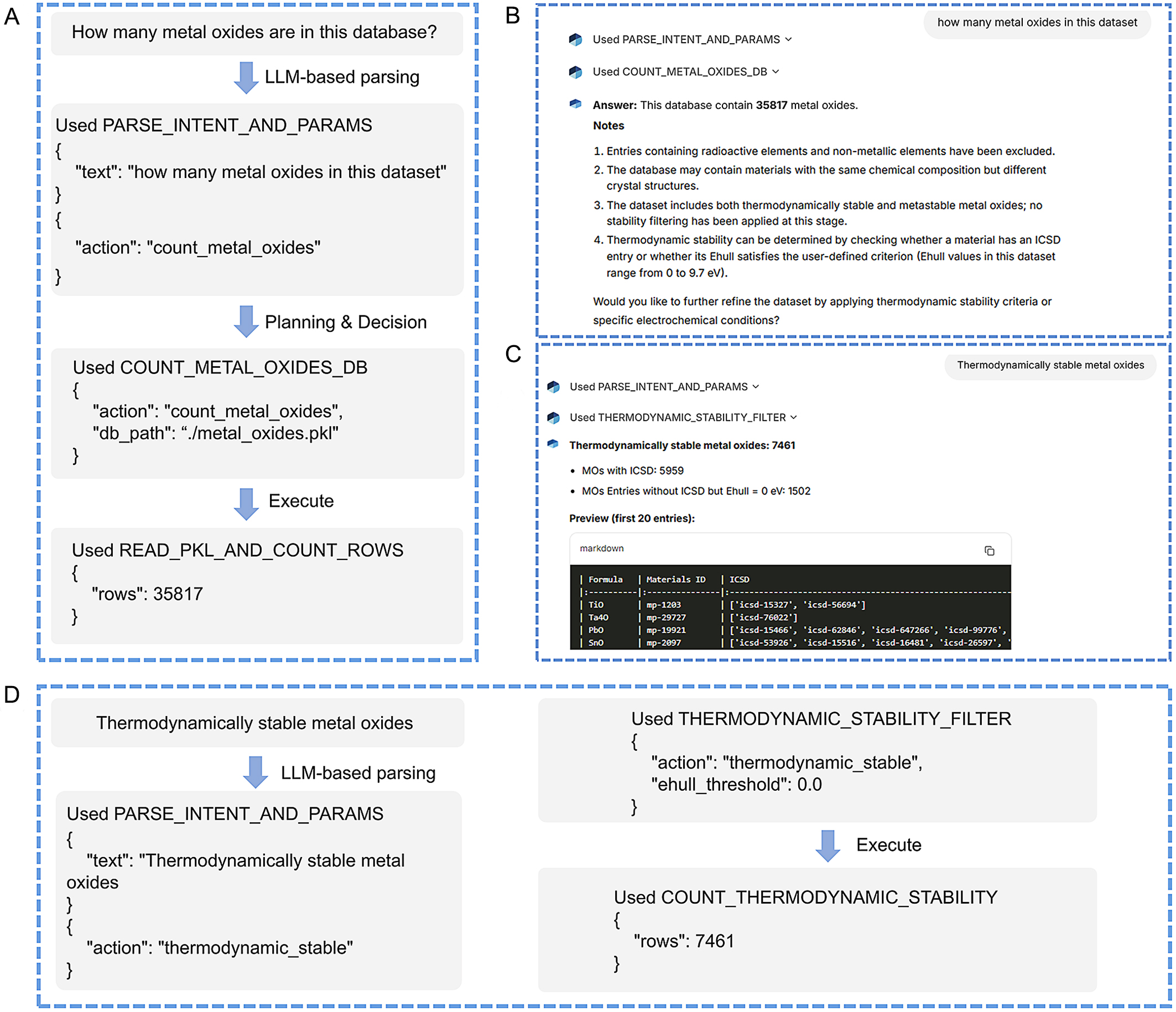
Figure 3 presents representative execution traces of the Agent in response to natural-language queries that quantitatively analyze the total number of MOs and their thermodynamic stability, corresponding to Action 1 in Figure 2. Figure 3A shows how the Agent works to show the dataset size of MOs. When the user asks, “How many metal oxides are in this dataset?”, the query is first processed by the LLM-based intent and parameter parsing module. The Agent interprets the request and triggers the corresponding action, namely “count_metal_oxides”. The Agent then queries the underlying local database, executes the action, and returns a structured response reporting the total number of MOs (35,817) [Figure 3B]. Additional contextual information is automatically provided to enhance interpretability, including dataset curation rules (e.g., exclusion of radioactive elements), the presence of multiple crystal structures for the same composition, and the inclusion of both thermodynamically stable and metastable materials for subsequent screening tasks. Users can further refine their query to focus on thermodynamically stable MOs. The resulting output is presented in Figure 3C, including both the total count and a preview of the filtered data. Figure 3D illustrates the thermodynamic stability screening workflow implemented in StableOx-Cat, focusing on bulk phase stability. When the user queries thermodynamically stable MOs, the agent again employs the LLM-based intent and parameter parsing module to identify thermodynamic stability as the target task. It then plans and invokes a dedicated thermodynamic stability filtering pipeline. The Ehull threshold can be specified by the user within the range of 0-9.7 eV; if not specified, a default value of 0 eV is applied. Based on this criterion, the agent identifies 7,461 thermodynamically stable MOs, including 5,959 MOs with reported ICSD entries and 1,502 MOs without ICSD entries but with Ehull =0 eV, and then returns these results in a structured format as shown in Figure 3C. If the user provides a custom Ehull threshold, the agent uses it to identify stable materials.
Figure 3. (A) Representative examples of how the Agent works to show the dataset size of MOs; (B) Interface of the StableOx-Cat Agent showing the result of (A); (C) Interface of the StableOx-Cat Agent showing the result of (D) defining thermodynamic information. MOs: Metal oxides; ICSD: Inorganic Crystal Structure Database.
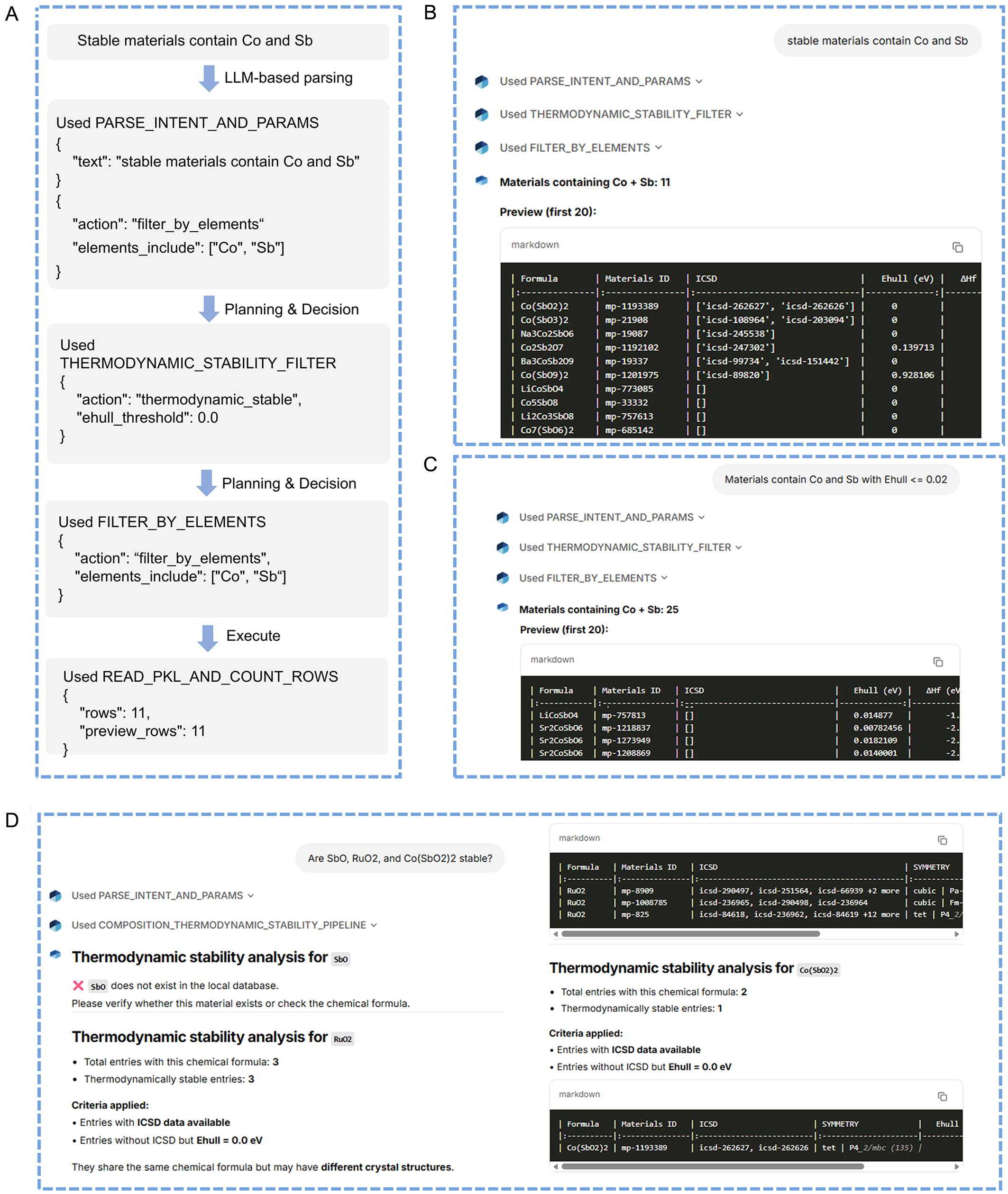
Figure 4 illustrates representative execution examples of the StableOx-Cat Agent for element-constrained thermodynamic stability screening and specific MOs stability verification corresponding to Actions 2 and 3 in Figure 2. Figure 4A presents the internal decision workflow triggered by the natural-language query “stable materials contain Co and Sb.” The query is first processed by the LLM-based intent and parameter parsing module to extract the action (filter_by_elements) and identify the required elements (Co and Sb). The agent then invokes the thermodynamic stability filter with a strict default criterion of Ehull = 0.0 eV, followed by “filter_by_elements” to retain MOs containing both Co and Sb. Following this workflow, the local database is queried to get all stable MOs containing Co and Sb, as shown in Figure 4B. The Agent reports the total number of MOs containing Co and Sb under strict thermodynamic stability criteria and provides a preview of the first 20 entries, including key metadata such as formula, Materials ID, ICSD information, Ehull, and formation energy. Figure 4C demonstrates a relaxed stability condition, where the user specifies Ehull ≤ 0.02 eV. The agent dynamically updates the thermodynamic filtering threshold, leading to a higher number of identified MOs. This highlights the system’s flexibility in handling user-defined stability criteria. Figure 4D presents the specific-composition stability verification [e.g., SbO, RuO2, and Co(SbO2)2]. For compositions not found in the local dataset, the agent provides a clear notification and prompts the user to verify the formula. For existing compositions, the agent reports the total number of entries, the number of thermodynamically stable entries, and the applied criteria (ICSD availability or Ehull = 0.0 eV). When multiple structures share the same chemical formula, the Agent explicitly indicates structural diversity, emphasizing phase-dependent stability.
Figure 4. (A) Representative examples of how the Agent works to show the user-defined elements contained in thermodynamically stable MOs and (B) the corresponding results shown in the interface. The interface of the StableOx-Cat Agent displays (C) the user-defined elements within a user-defined thermodynamic threshold and (D) specific-composition stability verification. LLM: large language model; ICSD: Inorganic Crystal Structure Database; ID: identifier.
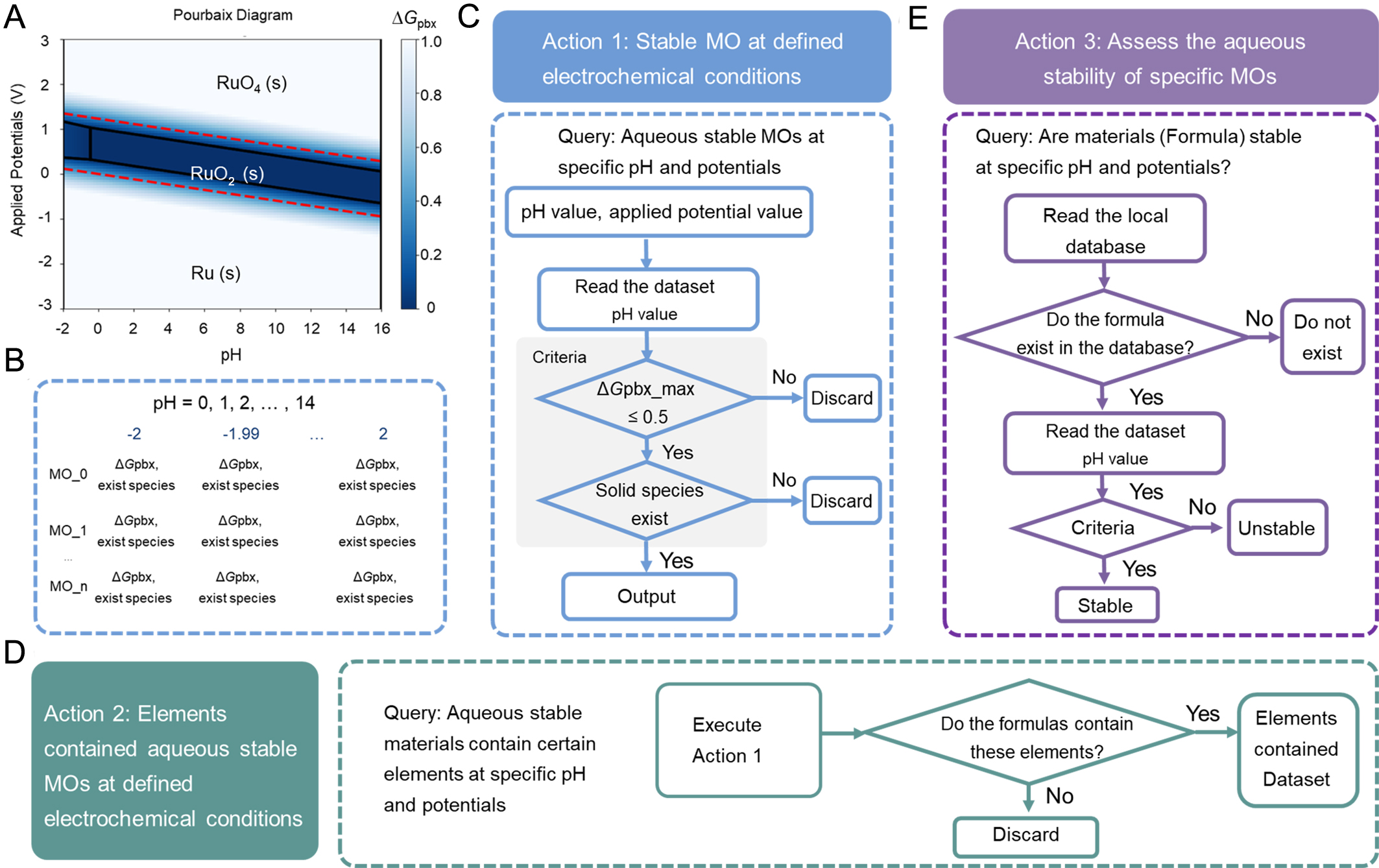
Figure 5 illustrates the construction of the aqueous stability evaluation framework and its integration into the Agent-based workflow. To clarify the physical insights for assessing aqueous stability under specific electrochemical conditions, we present a representative bulk Pourbaix diagram of RuO2, a well-known electrocatalyst [Figure 5A]. The diagram describes the ΔGpbx and the thermodynamically stable phases as functions of pH and applied potential (vs. SHE). A material is considered electrochemically stable at a defined pH-potential condition when ΔGpbx ≤ 0.5 eV·atom-1[16] and a solid-phase domain exists under those conditions. To enable systematic stability assessment, key information was extracted from bulk Pourbaix diagrams and stored in a structured local database, as shown in Figure 5B. For each pH condition, at 15 discrete pH values (pH = 0-14), ΔGpbx and the corresponding stable phase were recorded across the potential window from -2 to 2 V, yielding 15 structured datasets. Based on this database, three electrochemical-stability-related action modules were developed to support automated, condition-specific aqueous stability screening within the agent framework.
Figure 5. (A) A representative bulk Pourbaix diagram of RuO2; (B) The dataset information for the aqueous analysis. The functional architecture for the analysis of aqueous stable MOs, including (C) stable MOs analysis, (D) stable MOs containing specific elements analysis, and (E) assessing the aqueous stability for specific MOs at defined electrochemical conditions. MOs: Metal oxides.
Action 1 [Figure 5C] identifies all MOs that are stable under user-defined electrochemical conditions, specified by pH and applied potential. Based on the specified pH, the Agent selects the dataset corresponding to that pH from the pre-constructed database. The stability criteria are then applied: an MO is considered electrochemically stable at the specified condition if the maximum ΔGpbx satisfies ≤ 0.5 eV·atom-1[16] and a solid-phase domain exists[18,19] at the given potential. By applying these criteria, the agent retrieves all MOs that are stable under the defined electrochemical condition. Action 2 [Figure 5D] enables element-based statistical analysis of aqueous-stable MOs. After executing Action 1 with the defined pH and applied potentials, the agent further filters the electrochemically stable MOs based on user-defined elemental constraints and summarizes the filtered results. Action 3 [Figure 5E] performs stability assessment for specific user-defined MOs. The Agent first checks whether the queried MOs exist in the database. If present, it retrieves the corresponding data at the specified pH and evaluates its stability using the same criteria described above (ΔGpbx ≤ 0.5 eV·atom-1 and the presence of a solid-phase domain at the given potential). The Agent then reports whether the MO is electrochemically stable under the defined conditions.
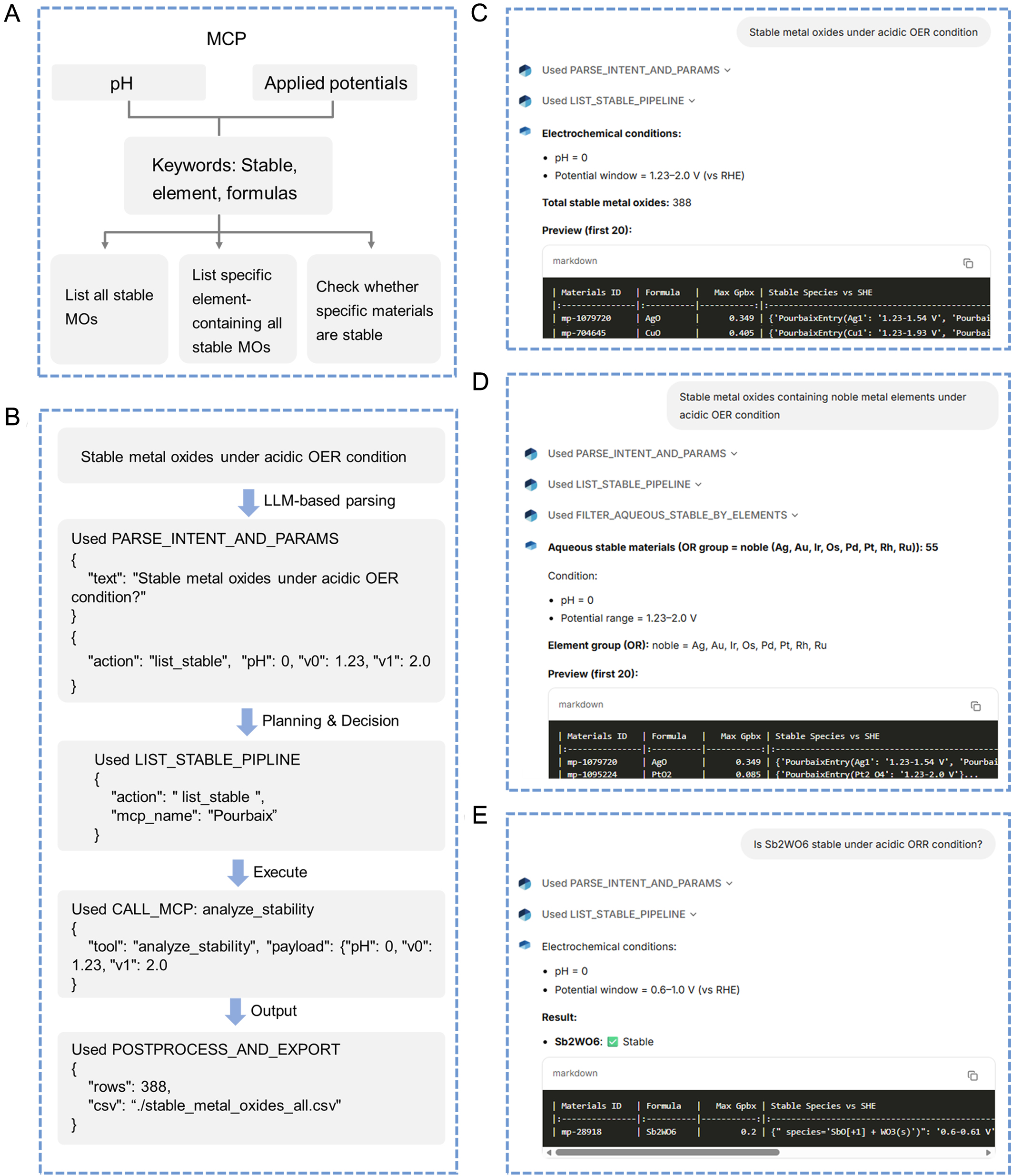
Figure 6 illustrates representative execution examples of the StableOx-Cat Agent for aqueous stability screening of MOs under specific electrochemical conditions corresponding to the actions defined in Figure 5. Figure 6A schematically presents the logical framework underlying the StableOx-Cat Agent, which is deployed as an MCP (details are provided in the METHODS section). User-defined electrochemical parameters, including pH and applied potential windows, are interpreted in conjunction with key information extracted by the LLM, such as “stable,” “specific elements”, or “material formulas”. Based on these structured signals, the Agent categorizes user queries into three primary task types (i.e., three actions in Figure 5): (i) listing all electrochemically stable MOs under specified conditions; (ii) retrieving stable MOs containing particular elements or element groups; and (iii) evaluating the stability of user-specified compounds. Figure 6B presents a representative workflow for querying electrochemically stable MOs under defined aqueous conditions. Upon receiving a natural language request (e.g., “Stable metal oxides under acidic OER condition”), the Agent first invokes the parsing module to extract structured parameters, including pH and the applied potential window. The resulting JSON (JavaScript Object Notation) output is then routed to the LIST_STABLE_PIPELINE, which calls the MCP backend (i.e., analyze_stability) to perform Pourbaix-based stability evaluation. The computed results are subsequently post-processed and formatted, including the total number of stable materials and a preview of representative entries, as shown in the interface in Figure 6B. Furthermore, when specific elements or material formulas are included in the query, the agent automatically triggers additional action modules, such as element-based filtering or compound stability verification, as illustrated in Figure 6C and D. Therefore, our agent can effectively determine stable MOs under user-defined electrochemical conditions. It should be noted that bulk Pourbaix diagrams provide an efficient thermodynamic framework for rapidly identifying MOs that remain stable within defined potential and pH ranges. However, the stability predicted from bulk thermodynamics does not necessarily represent the actual catalyst surface under operating electrochemical conditions. In practice, surface structures can be modified by the adsorption of reaction intermediates (e.g., HO*, O*, and H*) or by the formation of oxygen vacancies during electrochemical water activation[25-27]. Furthermore, surface configurations can evolve with changes in pH, resulting in distinct catalytic performance[28]. Therefore, while bulk Pourbaix screening provides an effective first step for narrowing the materials space, incorporating surface states and their pH-dependent evolution is critical for the reliable evaluation of catalyst stability and activity.
Figure 6. (A) The logical framework underlying the StableOx-Cat Agent, which is deployed as an MCP; (B) Representative examples of how the agent works to show the results of aqueous stable MOs at a specific electrochemical reaction, and (C) the interface of the agent results of (B). The agent interface results for (D) user-defined-element stable MOs and (E) specific-composition stability verification for a specific electrochemical reaction. MCP: Model Context Protocol; MOs: metal oxides; OER: oxygen evolution reaction; LLM: large language model; RHE: reversible hydrogen electrode; ORR: oxygen reduction reaction.
CONCLUSIONS
In this work, we developed StableOx-Cat, an AI agent framework designed to facilitate the systematic exploration of MO electrocatalysts through a natural-language-driven interface. By integrating LLM-based intent parsing with deterministic physics-based stability evaluation, StableOx-Cat enables automated analysis of both bulk thermodynamic stability and aqueous electrochemical stability under user-defined conditions. The framework combines structured database information with well-defined stability criteria, providing a reproducible and efficient workflow for stability-oriented catalyst screening. Overall, StableOx-Cat lowers the technical barrier to stability-oriented materials analysis and provides a flexible, extensible platform for identifying experimentally relevant MO electrocatalysts. The Agent-based architecture can be readily adapted to other electrocatalyst systems, electrochemical reactions, and data-driven discovery tasks.
DECLARATIONS
Authors’ contributions
Investigation and writing - original draft: Jia, X.
Data analysis and methodology: Jia, X.; Zhang, D.; Lu, Y.; Wang, Q.
Conceptualization: Jia, X.; Li, H.
Writing - review and editing, supervision, and funding acquisition: Li, H.
Availability of data and materials
The source code for StableOx-Cat is publicly available at: https://github.com/XueJiaAIMR/StableOx-Cat-Agent. Relevant Video Demo can be found in the Supplementary Materials.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
This work was supported by JSPS KAKENHI (Nos. JP25H01508, JP25K01737, JP25K17991, and JP24K23068).
Conflicts of interest
Li, H. is the Editor-in-Chief of AI Agent. Li, H. was not involved in any steps of the editorial process, including reviewers’ selection, manuscript handling, or decision-making. The other authors declare that there are no conflicts of interest.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
Supplementary Materials
REFERENCES
1. Li, H.; Kelly, S.; Guevarra, D.; et al. Analysis of the limitations in the oxygen reduction activity of transition metal oxide surfaces. Nat. Catal. 2021, 4, 463-8.
2. Zhu, Y.; Tang, Z.; Yuan, L.; Li, B.; Shao, Z.; Guo, W. Beyond conventional structures: emerging complex metal oxides for efficient oxygen and hydrogen electrocatalysis. Chem. Soc. Rev. 2025, 54, 1027-92.
3. Liu, H.; Sun, X.; Gao, F.; Zheng, Y.; Qiao, S. Cost-efficient and stable electrolysis of reverse osmosis water using a Co-RuO2-enabled PEM electrolyser. Nat. Catal. 2026, 9, 9-17.
4. Qin, Y.; Yu, T.; Deng, S.; et al. RuO2 electronic structure and lattice strain dual engineering for enhanced acidic oxygen evolution reaction performance. Nat. Commun. 2022, 13, 3784.
5. Song, F.; Bai, L.; Moysiadou, A.; et al. Transition metal oxides as electrocatalysts for the oxygen evolution reaction in alkaline solutions: an application-inspired renaissance. J. Am. Chem. Soc. 2018, 140, 7748-59.
6. Jain, A.; Ong, S. P.; Hautier, G.; et al. Commentary: The Materials Project: a materials genome approach to accelerating materials innovation. APL. Mater. 2013, 1, 011002.
7. Curtarolo, S.; Setyawan, W.; Hart, G. L.; et al. AFLOW: an automatic framework for high-throughput materials discovery. Comput. Mater. Sci. 2012, 58, 218-26.
8. Zagorac, D.; Müller, H.; Ruehl, S.; Zagorac, J.; Rehme, S. Recent developments in the Inorganic Crystal Structure Database: theoretical crystal structure data and related features. J. Appl. Crystallogr. 2019, 52, 918-25.
9. Zhang, D.; Li, H. Digital Catalysis Platform (DigCat): a gateway to big data and AI-powered innovations in catalysis. ChemRxiv 2024. Available online: https://doi.org/10.26434/chemrxiv-2024-9lpb9 (accessed 23 March 2026).
10. Ong, S. P.; Cholia, S.; Jain, A.; et al. The Materials Application Programming Interface (API): a simple, flexible and efficient API for materials data based on REpresentational State Transfer (REST) principles. Comput. Mater. Sci. 2015, 97, 209-15.
11. Ong, S. P.; Richards, W. D.; Jain, A.; et al. Python Materials Genomics (pymatgen): a robust, open-source python library for materials analysis. Comput. Mater. Sci. 2013, 68, 314-9.
12. Aykol, M.; Dwaraknath, S. S.; Sun, W.; Persson, K. A. Thermodynamic limit for synthesis of metastable inorganic materials. Sci. Adv. 2018, 4, eaaq0148.
13. Jain, A.; Hautier, G.; Ong, S. P.; et al. Formation enthalpies by mixing GGA and GGA + U calculations. Phys. Rev. B. 2011, 84, 045115.
14. Wang, A.; Kingsbury, R.; McDermott, M.; et al. A framework for quantifying uncertainty in DFT energy corrections. Sci. Rep. 2021, 11, 15496.
15. Persson, K. A.; Waldwick, B.; Lazic, P.; Ceder, G. Prediction of solid-aqueous equilibria: Scheme to combine first-principles calculations of solids with experimental aqueous states. Phys. Rev. B. 2012, 85, 235438.
16. Singh, A. K.; Zhou, L.; Shinde, A.; et al. Electrochemical stability of metastable materials. Chem. Mater. 2017, 29, 10159-67.
17. Patel, A. M.; Nørskov, J. K.; Persson, K. A.; Montoya, J. H. Efficient Pourbaix diagrams of many-element compounds. Phys. Chem. Chem. Phys. 2019, 21, 25323-7.
18. Jia, X.; Yu, Z.; Liu, F.; et al. Identifying stable electrocatalysts initialized by data mining: Sb2WO6 for oxygen reduction. Adv. Sci. 2024, 11, e2305630.
19. Jia, X.; Zhou, Z.; Liu, F.; et al. Closed-loop framework for discovering stable and low-cost bifunctional metal oxide catalysts for efficient electrocatalytic water splitting in acid. J. Am. Chem. Soc. 2025, 147, 22642-54.
20. Li, H. AI-driven multi-agent collaborations for accelerating catalyst design. Natl. Sci. Rev. 2026, 13, nwag067.
21. Zhang, D.; Jia, X.; Tran, H. B.; et al. “DIVE” into hydrogen storage materials discovery with AI agents. Chem. Sci. 2026, 17, 3031-42.
22. Kang, Y.; Kim, J. ChatMOF: an artificial intelligence system for predicting and generating metal-organic frameworks using large language models. Nat. Commun. 2024, 15, 4705.
23. Wang, Q.; Yang, F.; Wang, Y.; et al. Unraveling the complexity of divalent hydride electrolytes in solid-state batteries via a data-driven framework with large language model. Angew. Chem. Int. Ed. Engl. 2025, 64, e202506573.
24. Wang, Z.; Zheng, Y.; Chorkendorff, I.; Nørskov, J. K. Acid-stable oxides for oxygen electrocatalysis. ACS. Energy. Lett. 2020, 5, 2905-8.
25. Liu, H.; Jia, X.; Cao, A.; Wei, L.; D’agostino, C.; Li, H. The surface states of transition metal X-ides under electrocatalytic conditions. J. Chem. Phys. 2023, 158, 124705.
26. Liu, H.; Zhang, D.; Holmes, S. M.; D’Agostino, C.; Li, H. Origin of the superior oxygen reduction activity of zirconium nitride in alkaline media. Chem. Sci. 2023, 14, 9000-9.
27. Wang, T.; Guo, Z.; Oka, H.; Kumatani, A.; Liu, C.; Li, H. Origin of electrocatalytic nitrogen reduction activity over transition metal disulfides: critical role of in situ generation of S vacancy. J. Mater. Chem. A. 2024, 12, 8438-46.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data












Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.